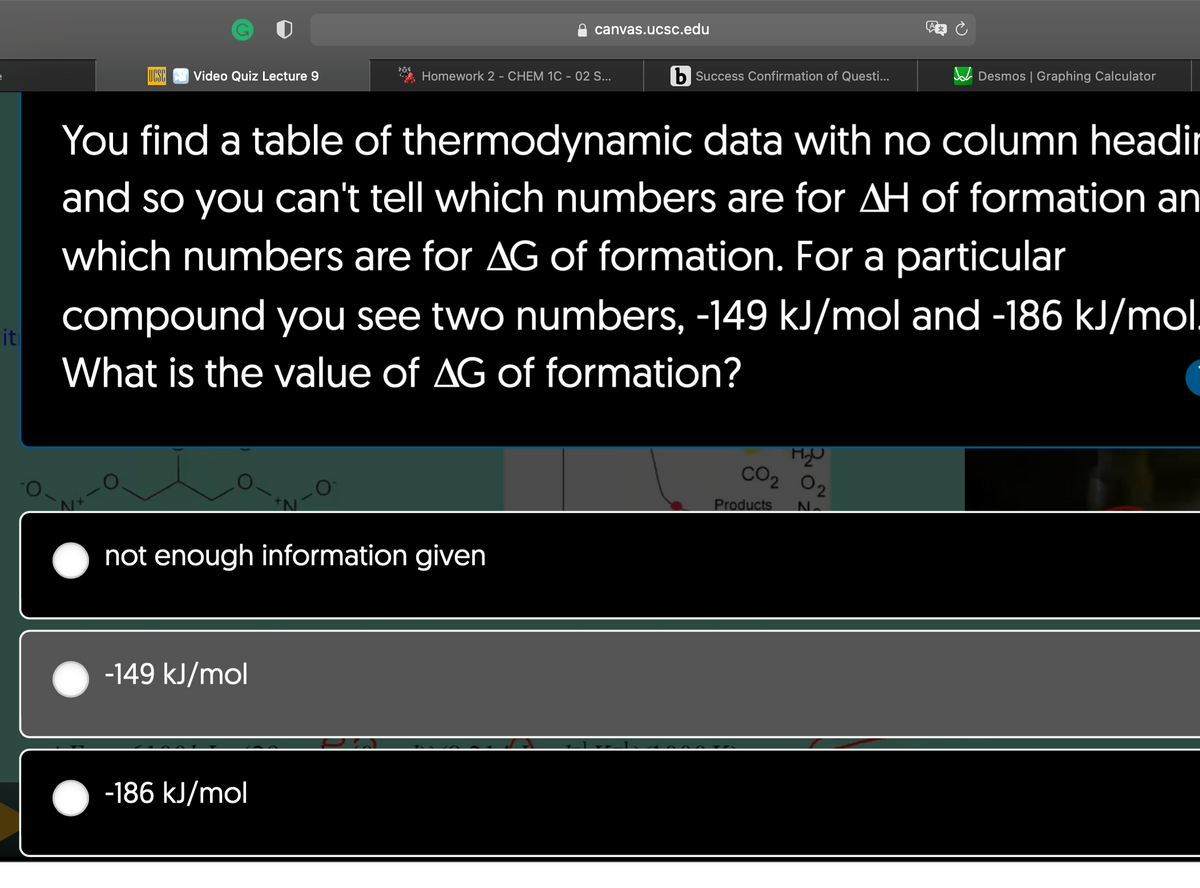
You find a table of thermodynamic data with no column headi and so you can't tell which numbers are for AH of formation ar which numbers are for AG of formation. For a particular compound you see two numbers, -149 kJ/mol and -186 kJ/mo What is the value of AG of formation?
You find a table of thermodynamic data with no column headi and so you can't tell which numbers are for AH of formation ar which numbers are for AG of formation. For a particular compound you see two numbers, -149 kJ/mol and -186 kJ/mo What is the value of AG of formation?
Chapter31: Introduction To Analytical Separations
Section: Chapter Questions
Problem 31.33QAP
Related questions
Question
Transcribed Image Text:it
UCSC Video Quiz Lecture 9
N+
284
-149 kJ/mol
not enough information given
-186 kJ/mol
canvas.ucsc.edu
Homework 2 - CHEM 1C - 02 S...
You find a table of thermodynamic data with no column headir
and so you can't tell which numbers are for AH of formation an
which numbers are for AG of formation. For a particular
compound you see two numbers, -149 kJ/mol and -186 kJ/mol.
What is the value of AG of formation?
b Success Confirmation of Questi...
H₂O
CO₂ 0₂
Desmos | Graphing Calculator
Products N.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

