
Concept explainers
The six elements in blue at the far right of the periodic table are gases at room temperature. The red elements in the middle of the table are the so-called coinage metals. Identify each of these elements using the periodic table inside the front cover of this book.
Interpretation:
The boxes that are highlighted red in colour has to be identified from the periodic table.
Concept Introduction:
Periodic table: It is arrangement of the chemical elements, ordered by their atomic number, electronic configuration and recurring chemical properties, whose structure shows periodic trends, than all elements present usually in rows, so that elements with similar atomic structure appear in vertical columns.
Answer to Problem 1.26UKC
The blue color indicated are Noble Gas family and the elements are Helium
Red color indicated in periodic table is the coinage metals namely, copper
Explanation of Solution
Let us consider the given statement of periodic table.
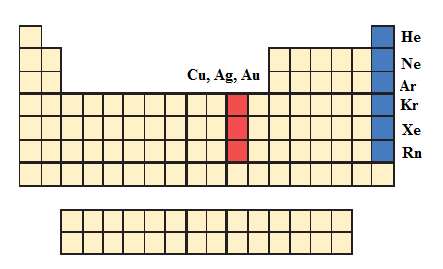
Above periodic table, and corresponding blue color indicated by Noble gas elements like Helium
Red color indicated in the periodic table is the coinage metals namely, copper
The elements were identified given the periodic table with respective colors.
Want to see more full solutions like this?
Chapter 1 Solutions
Fundamentals of General, Organic, and Biological Chemistry Plus Mastering Chemistry with Pearson eText -- Access Card Package (8th Edition)
Additional Science Textbook Solutions
Chemistry: A Molecular Approach
Brock Biology of Microorganisms (15th Edition)
University Physics with Modern Physics (14th Edition)
Chemistry: The Central Science (14th Edition)
Conceptual Physics (12th Edition)
Organic Chemistry (9th Edition)
- Polonium is a rare element with 33 radioisotopes. The most common one, 210Po, has 82 protons and 128 neutrons. When 210Po decays, it emits an alpha particle, which is a helium nucleus (2 protons and 2 neutrons). 210Po decay is tricky to detect because alpha particles do not carry very much energy compared to other forms of radiation. For example, they can be stopped by a single sheet of paper or a few inches of air. That is one reason that authorities failed to discover toxic amounts of 210Po in the body of former KGB agent Alexander Litvinenko until after he died suddenly and mysteriously in 2006. What element does an atom of 210Po become after it emits an alpha particle?arrow_forwardAn isotope of sodium (Na) has a mass number of 22. How many neutrons does it have? a. 11 b. 12 c. 22 d. 44arrow_forwardThe atomic number on an element tells us the number ofarrow_forward
- Atoms of the same element that differ only in the number of neutrons are known asarrow_forwarda certain element consists of two stable isotopes. the first has a mass of 138 amuarrow_forwardAn ion has 15 protons, 16 neutrons, and 18 electrons. The symbol for the ion is 31P3+. 31P3-. 31Ar3+. 34S+. 34S-.arrow_forward
- How can you tell in an IR spectrum if the compound has just one or two oxygens? What is the difference between them? Thank you!arrow_forwardRefer to the following figure (first three rows of the periodic table) to answer the questions below. What element does not react with other elements? Group of answer choices A. hydrogen B. helium C. oxygen D. siliconarrow_forwardWhat three things did the bohr model add to our understanding of atomic structure?arrow_forward
- The liquids butan-1-ol and butanal have similar molar masses. Which is expected to have the higher boiling point? Explain your choices.arrow_forwardWrite the nuclear equation for the positron decay of C-11.arrow_forwardThe atomic mass number of an atom is determined by the sum of the number of in the atom. (a) protons plus electrons plus neutrons (b) protons plus electrons (c) neutrons plus electrons (d) neutrons plus protonsarrow_forward

 Principles Of Radiographic Imaging: An Art And A ...Health & NutritionISBN:9781337711067Author:Richard R. Carlton, Arlene M. Adler, Vesna BalacPublisher:Cengage Learning
Principles Of Radiographic Imaging: An Art And A ...Health & NutritionISBN:9781337711067Author:Richard R. Carlton, Arlene M. Adler, Vesna BalacPublisher:Cengage Learning Biology Today and Tomorrow without Physiology (Mi...BiologyISBN:9781305117396Author:Cecie Starr, Christine Evers, Lisa StarrPublisher:Cengage Learning
Biology Today and Tomorrow without Physiology (Mi...BiologyISBN:9781305117396Author:Cecie Starr, Christine Evers, Lisa StarrPublisher:Cengage Learning Concepts of BiologyBiologyISBN:9781938168116Author:Samantha Fowler, Rebecca Roush, James WisePublisher:OpenStax College
Concepts of BiologyBiologyISBN:9781938168116Author:Samantha Fowler, Rebecca Roush, James WisePublisher:OpenStax College




