
Concept explainers
Interpretation:
To draw the structure of halothane, CF3CHCIBr, in three dimensions, using solid lines, wedges, dashes and to illustrate the position of atoms.
Concept introduction:
In the stereochemistry, various methods are depicted to represent three dimensional representations. Methane, a tetrahedral model, substituted by four different or same types of ligands at the vertices of the tetrahedron, which is not apparent in the
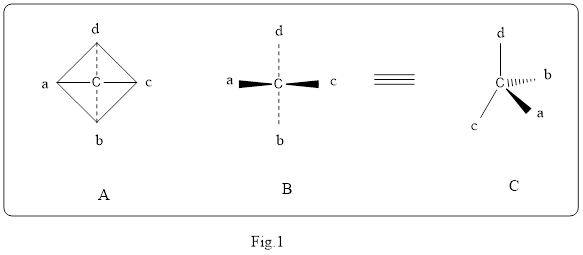
Wedge or solid heavy line represent a bond that project above the plane or pointing towards the observer. Broken wedge or dashed lines are that project below the plane or pointing away from the observer. The wedge and dashed lines for the sample methane is shown below in the Fig.1. Structure A and B represent a normal structure. Structure C represent the wedge and dashed lines. In the Structure C, a represent wedge, b represent dashed lines and c, d represent solid lines.
Want to see the full answer?
Check out a sample textbook solution
Chapter 1 Solutions
Organic Chemistry-Package(Custom)
- Draw and upload a Lewis structure for CH4. as well as answer the following questions. -what is the hybrizatjon around the carbon? - what is the electron pair geometry for your drawing? -what is the bond angle between the C-H bonds of CH4 what is the molecular shape for CH4?arrow_forwardDraw the structure of CH4, COH4, COH2, AND CO2H2arrow_forward(a) Draw four compounds with molecular formula C6H12O, each containing at least one different functional group, (b) Predict which compound has the highest boiling point, and explain your reasoning.arrow_forward
- Depict H-bonding between the molecule CH3OCH3 and waterarrow_forwardWhat is the accurate value of the CCH bond angle in ethylene (C2H4)? Please draw out the bond angles and explain your work.arrow_forwardA3 Why can ethylene glycol, HO-CH2CH2-OH, be used with water as an antifreeze in an automobile, but diethyl ether,CH3CH2OCH2CH3 , cannot? Explain in terms of polar and non-polar, particularly noting the ration of O atoms to C atoms for each compound.arrow_forward
- Draw a line-bond structure for propane, CH3CH2CH3. Predict the value of each bond angle, and indicate the overall shape of the molecule.arrow_forwardConsider the compound C₂H₃N. Which one of the structures in Figure 4 is the best representation of this compound based on your current knowledge? * A B C D All these structures are good representations of the compound.arrow_forwardThere are three different substances that contain a carbon-carbon double bond and have the formula C4H8. Draw them and tell how they differ.arrow_forward
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning

