
Concept explainers
(a)
Interpretation: The orbitals used to form the highlighted bonds are to be predicted.
Concept introduction: Hybridization is the combination of two or more atomic orbitals to form the same number of hybrid orbitals, each having the same shape and energy.
Answer to Problem 1.65P
Orbitals used in the formation of given bonds are:
Explanation of Solution
The given compound is,
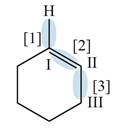
Figure
In bond
Orbitals used in the formation of given bonds are:
(b)
Interpretation: The orbitals used to form the highlighted bonds are to be predicted.
Concept introduction: Hybridization is the combination of two or more atomic orbitals to form the same number of hybrid orbitals, each having the same shape and energy.
Answer to Problem 1.65P
Orbitals used in the formation of given bonds are:
Explanation of Solution
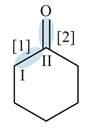
Figure 2
In bond
Orbitals used in the formation of given bonds are:
(c)
Interpretation: The orbitals used to form the highlighted bonds are to be predicted.
Concept introduction: Hybridization is the combination of two or more atomic orbitals to form the same number of hybrid orbitals, each having the same shape and energy.
Answer to Problem 1.65P
Orbitals used in the formation of given bonds are:
Explanation of Solution
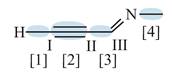
Figure 3
In bond
(a)
Want to see more full solutions like this?
Chapter 1 Solutions
Loose Leaf for Organic Chemistry
- Determine the geometry around the highlighted atom in each species ?arrow_forwardPlease help me out, indicate bond angles at the highlighted positions.arrow_forwardDescribe the orbitals used by each carbon atom in bonding and indicate the approximate bond angles. Drag the appropriate items to their respective bins.arrow_forward
- Label each carbon atom with the appropriate hybridizationarrow_forwardWhich of the given resonance structures (A, B,or C) contributes most to the resonance hybrid? Which contributes least?arrow_forwardFor parts A, B, and C label each carbon in the strucutres with the correct hybridization.arrow_forward
- Draw the corresponding resonance hybrid for the three structures.arrow_forwardProvide the two resonance structures of the molecules shown. Circle the best resonance structure out of the two.arrow_forwardDraw using the correct orbital overlap CO. Indicate the orbitals used for each bond formed.arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
