
ORGANIC CHEMISTRY W/CONNECT PKG
5th Edition
ISBN: 9781260901269
Author: SMITH
Publisher: MCG CUSTOM
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 1, Problem 1.72P
Anacin is an over-the-counter pain reliever that contains aspirin and caffeine. Answer the following
questions about each compound.
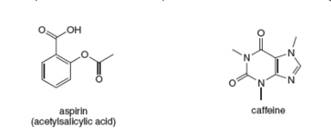
a. What is the molecular formula?
b. How many lone pairs are present on heteroatoms?
c. Label the hybridisation state of each carbon.
d. Draw three additional resonance structures.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Use the ball-and-stick model to answer each question about carbidopa, a drug used in combination with L-dopa to treat Parkinson's disease.a.Draw a skeletal structure of carbidopa.
b. Determine the hybridization around each carbon atom.c. What is the hybridization and geometry around each N atom?
d.How many polar bonds are present?
a. What is the hybridization of each N atom in nicotine?
b. What is the geometry around each N atom?
c. In what type of orbital does the lone pair on each N atom reside?
d. Draw a constitutional isomer of nicotine.
e. Draw a resonance structure of nicotine.
Anacin is an over-the-counter pain reliever that contains aspirin and caffeine. Answer the following questions about each compound.
a.What is the molecular formula?
b. How many lone pairs are present on heteroatoms?
c. Label the hybridization state of each carbon.
d. Draw three additional resonance structures.
Chapter 1 Solutions
ORGANIC CHEMISTRY W/CONNECT PKG
Ch. 1 - While the most common isotope of nitrogen has a...Ch. 1 - Label each bond in the following compounds as...Ch. 1 - How many covalent bonds are predicted for each...Ch. 1 - Draw a valid Lewis structure for each species. a....Ch. 1 - Draw an acceptable Lewis structure for each...Ch. 1 - Prob. 1.6PCh. 1 - Draw a Lewis structure for each ion. a. CH3Ob....Ch. 1 - Prob. 1.8PCh. 1 - Draw Lewis structures for each molecular formula....Ch. 1 - Classify each pair of compounds as isomers or...
Ch. 1 - Prob. 1.11PCh. 1 - Prob. 1.12PCh. 1 - Prob. 1.13PCh. 1 - Draw a second resonance structure for each species...Ch. 1 - Prob. 1.15PCh. 1 - Prob. 1.16PCh. 1 - Prob. 1.17PCh. 1 - Prob. 1.18PCh. 1 - Using the principles of VSEPR theory, you can...Ch. 1 - Convert each condensed formula to a Lewis...Ch. 1 - Prob. 1.21PCh. 1 - Prob. 1.22PCh. 1 - Convert each skeletal structure to a complete...Ch. 1 - What is the molecular formula of quinine, the...Ch. 1 - Draw in all hydrogens and lone pairs on the...Ch. 1 - Prob. 1.26PCh. 1 - What orbitals are used to form each of the CC, and...Ch. 1 - What orbitals are used to form each bond in the...Ch. 1 - Determine the hybridization around the highlighted...Ch. 1 - The unmistakable odor of a freshly cut cucumber is...Ch. 1 - Prob. 1.31PCh. 1 - Rank the following atoms in order of increasing...Ch. 1 - Prob. 1.33PCh. 1 - Prob. 1.34PCh. 1 - Provide the following information about...Ch. 1 - Use the ball-and-stick model to answer each...Ch. 1 - Citric acid is responsible for the tartness of...Ch. 1 - Zingerone gives ginger its pungent taste. a.What...Ch. 1 - Assign formal charges to each carbon atom in the...Ch. 1 - Assign formal charges to each and atom in the...Ch. 1 - Prob. 1.41PCh. 1 - Prob. 1.42PCh. 1 - Prob. 1.43PCh. 1 - Draw all possible isomers for each molecular...Ch. 1 - 1.45 Draw Lewis structures for the nine isomers...Ch. 1 - Prob. 1.46PCh. 1 - Prob. 1.47PCh. 1 - Prob. 1.48PCh. 1 - Prob. 1.49PCh. 1 - Prob. 1.50PCh. 1 - Prob. 1.51PCh. 1 - Prob. 1.52PCh. 1 - Consider compounds A-D, which contain both a...Ch. 1 - Prob. 1.54PCh. 1 - Prob. 1.55PCh. 1 - 1.56 Consider the compounds and ions with curved...Ch. 1 - 1.57 Predict all bond angles in each...Ch. 1 - 1.58 Predict the geometry around each highlighted...Ch. 1 - Prob. 1.59PCh. 1 - Draw in all the carbon and hydrogen atoms in each...Ch. 1 - 1.61 Convert each molecule into a skeletal...Ch. 1 - Prob. 1.62PCh. 1 - Prob. 1.63PCh. 1 - Predict the hybridization and geometry around each...Ch. 1 - Prob. 1.65PCh. 1 - Ketene, , is an unusual organic molecule that has...Ch. 1 - Rank the following bonds in order of increasing...Ch. 1 - Prob. 1.68PCh. 1 - Two useful organic compounds that contain Cl atoms...Ch. 1 - Use the symbols + and to indicate the polarity of...Ch. 1 - Prob. 1.71PCh. 1 - Anacin is an over-the-counter pain reliever that...Ch. 1 - Answer the following questions about acetonitrile...Ch. 1 - Prob. 1.74PCh. 1 - 1.75 The principles of this chapter can be...Ch. 1 -
a. What is the hybridization of each N atom in...Ch. 1 - 1.77 Stalevo is the trade name for a medication...Ch. 1 - 1.78 and are two highly reactive carbon...Ch. 1 - 1.79 The N atom in (acetamide) is hybridized,...Ch. 1 - Prob. 1.80PCh. 1 - Prob. 1.81PCh. 1 - Prob. 1.82PCh. 1 - Prob. 1.83PCh. 1 - Prob. 1.84PCh. 1 - Prob. 1.85P
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Help! a.) Draw all the important resonance contributors for the structure shown below. Use curved arrows to show the movement of electrons between these contributors. b.) Draw the hybrid resonance hybrid structure.arrow_forwardRizatriptan (trade name Maxalt) is a prescription drug used for the treatment of migraines. (a) How many aromatic rings does rizatriptan contain? (b) Determine the hybridization of each N atom. (c) In what type of orbital does the lone pair on each N reside? (d) Draw all the resonance structures for rizatriptan that contain only neutral atoms. (e) Draw all reasonable resonance structures for the five-membered ring that contains three N atoms.arrow_forwardConsider the following orbital representation of HCCH (ethyne). a. Answer the same three questions (a-c) from the previous exercise. b. Label each CH bond in the drawing above. c. What is the total number of a bonds found in ethyne?…. bonds? d. How many p orbitals are there on a single carbon of ethyne? e. How many hybrid orbitals are there on a single carbon of ethyne?arrow_forward
- Explain why the two molecules below cannot inter-convert without breaking the portion of thedouble bond.arrow_forwardSee the Attachment & Answer the following question? Stalevo is the trade name for a medication used for Parkinson's disease,which contains L-dopa, carbidopa, and entacapone. Ques: Use curved arrows to draw a resonance structure that is a minorcontributor to the resonance hybrid ?arrow_forwardWhat orbitals are used to form each highlighted bond? For multiplebonds, indicate the orbitals used in individual bonds ?arrow_forward
- What orbitals are used to form each highlighted bond? For multiple bonds, indicate the orbitals used in individual bonds.arrow_forwardDraw a second resonance structure for each ion. Then, draw the resonance hybrid.arrow_forwardConsidering each of the given ball-and-stick models: [1] Does the dark red atom have one or more lone pairs of electrons on it? Explain your choice. [2] Give an example of a molecule that has the indicated geometry.arrow_forward
- How can you tell whether a lone pair is localized or delocalized?arrow_forwardTrue or False? Circle your answer.a. A resonance hybrid is a structure with equal contribution from each possible resonance structure.True Falseb. Localized electrons do not participate in resonance.True Falsearrow_forwarda.) How many regions of high electron density are around the central atom in sulfur tetrafluoride? b.) How many regions of high electron density are around the Carbon atom in: formic acid (HCOOH) ? methanol ? HCN ? carbon dioxide ? methane ?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning


Chemistry for Today: General, Organic, and Bioche...
Chemistry
ISBN:9781305960060
Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:Cengage Learning
Types of bonds; Author: Edspira;https://www.youtube.com/watch?v=Jj0V01Arebk;License: Standard YouTube License, CC-BY