
Concept explainers
The skeleton is benzaldoxime is
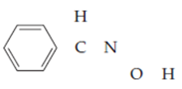
The number of valence electrons is as follows: from the phenyl group. ___; from each of two hydrogens, ___; from the carbon atom, ___; from the nitrogen atom, ___; and from the oxygen atom, ___, for a total of ___. Filling in the skeleton with single bonds and adding the appropriate unshared pairs gives
_____________________
The number of electrons in the functional group of this trial structure is ___, which is ___ too many. Removing an unshared pair from carbon and nitrogen and adding a second bond between them gives
_____________________
in which carbon, nitrogen, and oxygen have their customary valences of ___, ___, and respectively. The alternative structure with a double bond between nitrogen and oxygen is
_____________________
This structure is not acceptable because it requires carbon and oxygen to exhibit the unfamiliar valences of ___ and ___.
Derive Lewis structures for the compounds below.
Want to see the full answer?
Check out a sample textbook solution
Chapter 1 Solutions
Pushing Electrons
- Can you help me with the explanation of the alkane, alkene, alkyne, aromatic and all these terms give me examples too?arrow_forwardDuring our lectures on polymeric materials, I used polyethylene as an example because of its very straightforward structure. Draw a diagram of a long-chain molecule of polyethylene that consists of six units (or mers) of ethylene along with the starting and endpoint OH-. Include bonds in your diagram.arrow_forwardgive the nomenclature or the structure of the following molecules of item #21 to #25.arrow_forward
- The skeleton of benzyldimethylamine is The number of available valence electrons is as follows: from the benzyl group, ___; from each of two methyl groups, ___; and, from the nitrogen atom, ___, for a total of ___. Filling in the skeleton with single bonds gives ____________________________ and adding the appropriate unshared pair gives ____________________________ The number of electrons in the functional group of this structure is ___, and the structure is (correct, incorrect).arrow_forwardTo satisfy the octet rule, fullerenes must have doublebonds. How many? Give a simple rule for one way ofplacing them in the structure shown in Figure 7.17a.arrow_forwardName the following molecules, for which the structure is givenarrow_forward

 Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning

