
Concept explainers
Calculate the approximate number of atoms in a bacterium. Assume that the average mass of an atom in the bacterium is ten times the mass of a hydrogen atom. (Hint: The mass of a hydrogen atom is on the order of 10-27 kg and the mass of a bacterium is on the order of 10-15 kg.)
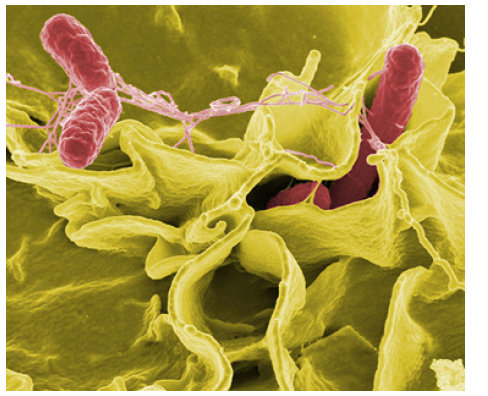
Figure 1.28 This color-enhanced photo shows Salmonella typhimurium (red) attacking human cells. These bacteria are commonly known for causing foodborne illness. Can you estimate the number of atoms in each bacterium? (credit: Rocky Mountain Laboratories, NIAID, NIH)
Trending nowThis is a popular solution!

Chapter 1 Solutions
College Physics
Additional Science Textbook Solutions
University Physics Volume 2
University Physics with Modern Physics (14th Edition)
College Physics: A Strategic Approach (4th Edition)
University Physics Volume 1
Physics for Scientists and Engineers: A Strategic Approach with Modern Physics (4th Edition)
Tutorials in Introductory Physics
- Roughly how many heartbeats are there in a lifetime?arrow_forwardIn general, when a derived unit becomes complicated (involves too many standard units), what is done?arrow_forwardA sidewalk is to be constructed around a swimming pool that measures (10.0 0.1) m by (17.0 0.1) m. If the sidewalk is to measure (1.00 0.01) m wide by (9.0 0.1) cm thick, what volume of concrete is needed and what is the approximate uncertainty of this volume?arrow_forward
- In a football game, you often hear the expression first and 10 (yards). How would you express this measurement in meters to a friend from Europe?arrow_forwardIf the DNA strand in a molecule could be stretched out, it would have a length on the order of 2.0 m. What would this be in feet and inches?arrow_forwardA generation is about one-third of a lifetime. Approximately how many generations have passed since the year 0 AD?arrow_forward
- Roughly how many times longer than the mean life of an extremely unstable atomic nucleus is the lifetime of a human?arrow_forwardA popular saying is Give him an inch, and hell take a mile. What would be the equivalent saying using comparable metric units?arrow_forwardOne student uses a meterstick to measure the thickness of a textbook and obtains 4.3 cm 0.1 cm. Other students measure the thickness with vernier calipers and obtain four different measurements: (a) 4.32 cm 0.01 cm. (b) 4.31 cm 0.01 cm, (c) 4.24 cm 0.01 cm, and (d) 4.43 cm 0.01 cm. Which of these four measurements, if any, agree with that obtained by the first student?arrow_forward
- Compute, in centimeters and in meters, the height of a basketball player who is 6 ft 5 in. tall.arrow_forwardIn SI units, speeds are measured in meters per second (m/s). But, depending on where you live, you’re probably mole comfortable of thinking of speeds in terms of either kilometers per hour (km/h) or miles per hour (mi/h). In this problem, you will see that 1 m/s is roughly 4 km/h or 2 mi/h, which is handy to use when developing your physical Intuition. More precisely, show that (a) 1.0m/s=3.6km/h and 1.0m/s=2.2mi/h .arrow_forwardA commonly used turn of mass in the English system Is the pound-mass, abbreviated Ibm, where 1 Ibm = 0.454 kg. What is the density of water in pound-mass per cubic foot?arrow_forward
 Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning University Physics Volume 1PhysicsISBN:9781938168277Author:William Moebs, Samuel J. Ling, Jeff SannyPublisher:OpenStax - Rice University
University Physics Volume 1PhysicsISBN:9781938168277Author:William Moebs, Samuel J. Ling, Jeff SannyPublisher:OpenStax - Rice University Physics for Scientists and Engineers: Foundations...PhysicsISBN:9781133939146Author:Katz, Debora M.Publisher:Cengage Learning
Physics for Scientists and Engineers: Foundations...PhysicsISBN:9781133939146Author:Katz, Debora M.Publisher:Cengage Learning An Introduction to Physical SciencePhysicsISBN:9781305079137Author:James Shipman, Jerry D. Wilson, Charles A. Higgins, Omar TorresPublisher:Cengage Learning
An Introduction to Physical SciencePhysicsISBN:9781305079137Author:James Shipman, Jerry D. Wilson, Charles A. Higgins, Omar TorresPublisher:Cengage Learning Glencoe Physics: Principles and Problems, Student...PhysicsISBN:9780078807213Author:Paul W. ZitzewitzPublisher:Glencoe/McGraw-Hill
Glencoe Physics: Principles and Problems, Student...PhysicsISBN:9780078807213Author:Paul W. ZitzewitzPublisher:Glencoe/McGraw-Hill College PhysicsPhysicsISBN:9781285737027Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning
College PhysicsPhysicsISBN:9781285737027Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning





