
Concept explainers
Ethanol,
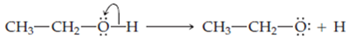
In this structure the oxygen owns one electron from
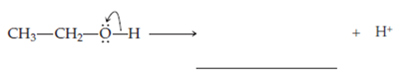
Note that the other fragment, the proton, leaves with a formal charge of +1.
Want to see the full answer?
Check out a sample textbook solution
Chapter 1 Solutions
Pushing Electrons
- Write Lewis structures for these ions. Show all valence electrons and all formal charges. (a) Amide ion, NH2 (b) Bicarbonate ion, HCO3 (c) Carbonate ion, CO32 (d) Nitrate ion, NO3 (e) Formate ion, HCOO (f) Acetate ion, CH3COOarrow_forwardThe n-propyl cation can be formed from a molecule such as When the C–Cl bond is broken so that both electrons leave with Cl, the fragments formed are The carbon atom that had been attached to Cl is now sharing ____ electron pairs. In each shared pair the carbon atom owns ____ electron. The number of electrons that belong to carbon is ____. The formal charge on the carbon atom is ____. The correct Lewis structure for the n-propyl cation isarrow_forwardDetermine the formal charge on each atom in the following molecules or ions: (a) SCO (b) HCO2 (formate ion) (c) CO32 (d) HCO2H (formic acid)arrow_forward
- The chemistry of the nitrite ion and HNO2: (a) Two resonance structures are possible for NO2. Draw these structures, and then find the formal charge on each atom in each resonance structure. (b) In forming the acid HNO2 an H+ ion attaches to the O atom and not the N atom of NO2. Explain why you would predict this result. (c) Two resonance structures are possible for HNO2. Draw these structures, and then find the formal charge on each atom in each resonance structure. Is either of these structures strongly preferred over the other?arrow_forwardWhat is the formal charge on the indicated atom in each of the following species? (a) sulfur in SO2 (b) nitrogen in N2H4 (c) each oxygen atom in ozone, O3arrow_forwardA common trait of simple organic compounds is to have Lewis structures where all atoms have a formal charge of zero. Consider the following incomplete Lewis structure for an organic compound called methyl cyanoacrylate, the main ingredient in Super Glue. Draw a complete Lewis structure for methyl cyanoacrylate in which all atoms have a formal charge of zero.arrow_forward
- Chloromethane has the Lewis structure _______________________________ The carbon atom is sharing 4 electron pairs. In each shared pair the carbon atom “owns” 1 electron. The number of electrons that “belong” to carbon is ___. Carbon, being a Group ___ element would have 4 , outer shell electrons in the unbonded, neutral state. Therefore, the carbon atom in chloromethane has a formal charge of zero.arrow_forwardCarbon and sulfur form compounds with each other with the formulas CS2 and C3S2. Draw a Lewis structure for each compound that has a formal charge of zero for all atoms in the structure.arrow_forwardFormamide, HC(O)NH2, is prepared at high pressures from carbon monoxide and ammonia, and serves as an industrial solvent (the parentheses around the O indicate that it is bonded only to the carbon atom and that the carbon atom is also bonded to the H and the N atoms). Two resonance forms (one with formal charges) can be written for formamide. Write both resonance structures, and predict the bond angles about the carbon and nitrogen atoms for each resonance form. Are they the same? Describe how the experimental determination of the HNH bond angle could be used to indicate which resonance form is more important.arrow_forward
- Aspartame is an artificial sweetener marketed under the name Nutra-Sweet. A partial Lewis structure for aspartame is shown below. Aspartame can be classified as an organic compound (a compound based on carbon atoms). The majority of Lewis structures for simple organic compounds have all atoms with a formal charge of zero. Therefore, add lone pairs and multiple bonds to the structure above to give each atom a formal charge of zero when drawing the Lewis structure. Also note that the six-sided ring is shorthand notation for a benzene ring (C6H5). Benzene is discussed in Section 4-7. Complete the Lewis structure for aspartame. How many C and N atoms exhibit sp1 hybridization? How many C and O atoms exhibit sp3 hybridization? How many and bonds are in aspartame?arrow_forwardConsider the NO 2 – ion. How many valence electrons does the ion have? How many lone pairs of electrons are on the central atom? How many lone pairs of electrons in total are on the substituent atoms? How many resonance structures can be drawn for the ion? If the ion doesn't exhibit resonance, indicate "1" as only one structure can be drawn for the ion.arrow_forward-draw the possible resonance structures of IO3^2- ion. -the experimental bond length of P and O bond in PO4^3- ion is in between the bond lengths of P=O and P-O.what is the reason behind this?. -draw the possible resonance structures of C3h3^- ion and on each sructure assign formal charge on each atom.arrow_forward

 Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning





