
Organic Chemistry: A Guided Inquiry
2nd Edition
ISBN: 9780618974122
Author: Andrei Straumanis
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 1, Problem 8E
a model of each of the following molecules: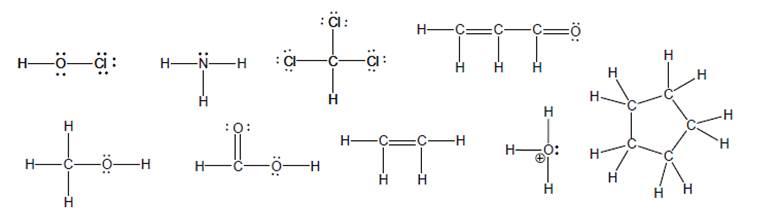
a. Based on your model, draw a bond-line representation with as many atoms as possible in the plane of the paper. Use wedge and dash bonds to represent any atoms that do not lie in the plane of the paper.
b. Indicate each unique bond angle and the shape of each unique central atom.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Draw an arrow to indicate each bond dipole. Circle any molecules that has a molecule dipole.
a) Draw a VSEPR diagram using the information below including all bond dipoles, the overall dipole, and any bond angles.
b) What is the name of the molecular shape/molecular geometry for your molecule?
c) Explain if your molecule is polar or non-polar.
Information:
Number of central atoms = 1
Number of other atoms surrounding the central atom = 4
Number of lone pairs surrounding the central atom = 1
The central atom is less electronegative than the surrounding atoms.
How important to draw the Lewis Diagram? Elaborate your answer.
Chapter 1 Solutions
Organic Chemistry: A Guided Inquiry
Ch. 1 - (E) What does the number (+Z) at the center of...Ch. 1 - Prob. 2CTQCh. 1 - Prob. 3CTQCh. 1 - Prob. 4CTQCh. 1 - Prob. 5CTQCh. 1 - Prob. 6CTQCh. 1 - Prob. 7CTQCh. 1 - You hear a student from a nearby group say that...Ch. 1 - Use VSEPR to explain why the HBH bond angle of BH3...Ch. 1 - Both the HCH and HCO bond angles of H2CO...
Ch. 1 - Prob. 11CTQCh. 1 - Consider the following flat drawing of methane...Ch. 1 - Use VSEPR to assign a value of (close to) 109.5,...Ch. 1 - A student draws the picture of ammonia (NH3) in...Ch. 1 - Prob. 15CTQCh. 1 - How many central atoms does the molecule H2NCH3...Ch. 1 - Indicate the bond angle and shape about each...Ch. 1 - Explain how there can be two kinds of bent:...Ch. 1 - A student makes the following statement: “The...Ch. 1 - A student who missed this class needs to know how...Ch. 1 - Prob. 1ECh. 1 - Prob. 2ECh. 1 - Consider the incomplete valence shell...Ch. 1 - How many valence electrons does a neutral a. K...Ch. 1 - Consider the molecules AlCl3 (aluminum chloride)...Ch. 1 - Draw an example of a bent molecule with a bond...Ch. 1 - Label each atom marked with an arrow with the...Ch. 1 - a model of each of the following molecules: a....
Additional Science Textbook Solutions
Find more solutions based on key concepts
Which of the following solutions has the higher molarity? 10 ppm KI in water or 10,000 ppb KBr in water 0.25 ma...
CHEMISTRY-TEXT
Real walls are never totally adiabatic. Use your experience to order the following walls in increasing order wi...
Thermodynamics, Statistical Thermodynamics, & Kinetics
4.1 Write the symbols for the following elements.
a. copper
b. platinum
c. calcium
d. manganese
e. Iron
...
Chemistry: An Introduction to General, Organic, and Biological Chemistry (13th Edition)
Practice Exercise 1
Which of the following factors determines the size of an atom? a. the volume of the nucleus...
Chemistry: The Central Science (14th Edition)
Draw a Lewis structure for each of the following species: a. H2CO3 b. CO32 c. CH2O d. CO2
Essential Organic Chemistry (3rd Edition)
Practice Exercise 1
Which of the following factors determines the size of an atom? a. the volume of the nucleus...
Chemistry: The Central Science (13th Edition)
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Part c draw the molecule by placing atoms on the grid and connecting them with bonds include all hydrogen atomsarrow_forwardFor a molecule to be polar it must have a difference in electronegativity between the atoms, a polar bond, and what else? Question 6 options: The exact location of the electron The probability of the electron being in that space The electronic orbital path. Nothing Shrodoedinger was just trying to entertain his daughter.arrow_forwardDraw a resonance structure that places a pi bond in a different position. Include all lone pairs in your structure. Show your work.arrow_forward
- Hydrogen bonds Group of answer choices A) Are the strongest individual bond within compounds B) describe the attraction of the positive hydrogen end of a polar molecule to the negative end of another polar molecule C) weaker at low temperatures D) describe the attraction of gases to each otherarrow_forwardDraw what happens as the approaching atoms begin to repel, slow down, and ultimately move apart.arrow_forwardVSEPR theory can be used to predict the shapes that simple molecules form. First, explain the concepts used in VSEPR theory to predict what shape a molecule with form. Then, using VSEPR theory, state the shapes that each of the following molecules will form.arrow_forward
- For this molecule : a) draw the resonance forms for this molecule and indicate the direction in which the electrons move. b) identify the most stable form and explain. c) draw the resonance hybrid.arrow_forwardProvide the molecular formula and Lewis structure, the expanded structural formula, for the hydrocarbon ball‑and‑stick model shown. Be sure to include all hydrogen atoms in your Lewis structure. Click on the three‑dimensional molecular structure of the compound and drag to rotate it, or use the controls provided. What is the molecular formula? Draw the lewis structurearrow_forwardTake a look at this molecule, and then answer the questions in the table below it.arrow_forward
- A)Draw the structure/geometry as predicted by the VSEPR theory b) Name the shape of the geometry as predicted by the VSEPR theory c) Indicate the bond angle(s) found in the particle d) Indicate the polarity of the particlearrow_forwardLabel three polar bonds ?arrow_forwarda. how many valence electrons are there? b. draw the valence bond energy diagram.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
 World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHER
Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHER

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:9781337399425
Author:Steven S. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning


World of Chemistry, 3rd edition
Chemistry
ISBN:9781133109655
Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:Brooks / Cole / Cengage Learning

Introductory Chemistry: An Active Learning Approa...
Chemistry
ISBN:9781305079250
Author:Mark S. Cracolice, Ed Peters
Publisher:Cengage Learning

Living By Chemistry: First Edition Textbook
Chemistry
ISBN:9781559539418
Author:Angelica Stacy
Publisher:MAC HIGHER
Stoichiometry - Chemistry for Massive Creatures: Crash Course Chemistry #6; Author: Crash Course;https://www.youtube.com/watch?v=UL1jmJaUkaQ;License: Standard YouTube License, CC-BY
Bonding (Ionic, Covalent & Metallic) - GCSE Chemistry; Author: Science Shorts;https://www.youtube.com/watch?v=p9MA6Od-zBA;License: Standard YouTube License, CC-BY
General Chemistry 1A. Lecture 12. Two Theories of Bonding.; Author: UCI Open;https://www.youtube.com/watch?v=dLTlL9Z1bh0;License: CC-BY