
Organic Chemistry
8th Edition
ISBN: 9781305580350
Author: William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 10, Problem 10.33P
Two diastereomeric sets of enantiomers, A/B and C/D, exist for 3-bromo-2-butanol.
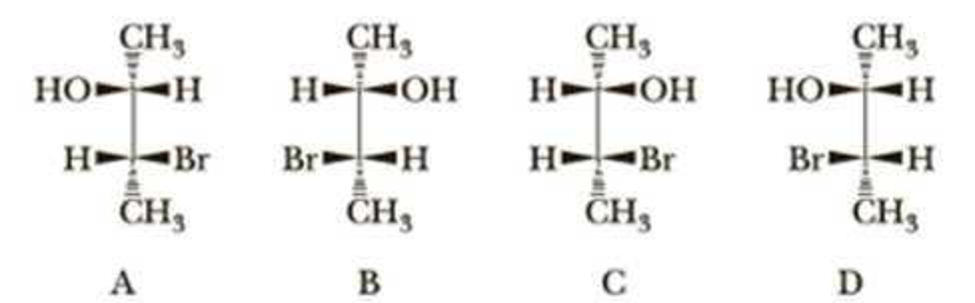
When enantiomer A or B is treated with HBr, only racemic 2,3-dibromobutane is formed; no meso isomer is formed. When enantiomer C or D is treated with HBr, only meso 2,3-dibromobutane is formed; no racemic 2,3-dibromobutane is formed. Account for these observations.
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
5-Hydroxyhexanal forms a six-membered cyclic hemiacetal, which predominates at equilibrium in aqueous solution.
(a) Draw a structural formula for this cyclic hemiacetal.
(b) How many stereoisomers are possible for 5-hydroxyhexanal?
(c) How many stereoisomers are possible for this cyclic hemiacetal?
(d) Draw alternative chair conformations for each stereoisomer and label groups axial or Also predict which of the alternative chair conformations for each stereoisomer is more stable.
Five isomeric alkanes (A–E) having the molecular formula C6H14 are each treated with Cl2 + hν to give alkyl halides having molecular formula C6H13Cl. A yields five constitutional isomers. B yields four constitutional isomers. C yields two constitutional isomers. D yields three constitutional isomers, two of which possess stereogenic centers. E yields three constitutional isomers, only one of which possesses a stereogenic center. Identify the structures of A–E.
Treatment of cis-4-bromocyclohexanol with HO− affords compound Aand cyclohex-3-en-1-ol. Treatment of trans-4-bromocyclohexanol under the same conditions forms compound B and cyclohex-3-en-1-ol. A and Bcontain different functional groups and are not isomers of each other.Propose structures for A and B and offer an explanation for theirformation.
Chapter 10 Solutions
Organic Chemistry
Ch. 10.1 - Write IUPAC names for these alcohols and include...Ch. 10.1 - Classify each alcohol as primary, secondary, or...Ch. 10.1 - Write IUPAC names for these unsaturated alcohols.Ch. 10.2 - Arrange these compounds in order of increasing...Ch. 10.2 - Prob. 10.5PCh. 10.4 - Predict the position of equilibrium for this...Ch. 10.5 - Show how to convert (R)-2-pentanol to...Ch. 10.6 - Draw structural formulas for the alkenes formed by...Ch. 10.6 - Propose a mechanism to account for this...Ch. 10.7 - Propose a mechanism to account for the following...
Ch. 10.7 - Prob. AQCh. 10.7 - Prob. BQCh. 10.7 - Prob. CQCh. 10.7 - Prob. DQCh. 10.7 - Which step in the reaction would you expect to be...Ch. 10.7 - Prob. FQCh. 10.7 - Prob. GQCh. 10.8 - Prob. 10.11PCh. 10.8 - Prob. AQCh. 10.8 - Prob. BQCh. 10.8 - Prob. CQCh. 10.8 - Why does nature use a reagent as complex as NAD+...Ch. 10.8 - -Hydroxyketones and -hydroxyaldehydes are also...Ch. 10.9 - Write IUPAC names for these thiols.Ch. 10 - Which are secondary alcohols?Ch. 10 - Name each compound.Ch. 10 - Prob. 10.16PCh. 10 - Name and draw structural formulas for the eight...Ch. 10 - Arrange these compounds in order of increasing...Ch. 10 - Arrange these compounds in order of increasing...Ch. 10 - Prob. 10.20PCh. 10 - Prob. 10.21PCh. 10 - Arrange the compounds in each set in order of...Ch. 10 - Prob. 10.23PCh. 10 - The decalinols A and B can be equilibrated using...Ch. 10 - Prob. 10.25PCh. 10 - Select the stronger acid from each pair and...Ch. 10 - Prob. 10.27PCh. 10 - In each equilibrium, label the stronger acid, the...Ch. 10 - Write equations for the reaction of 1-butanol with...Ch. 10 - Write equations for the reaction of 2-butanol with...Ch. 10 - Prob. 10.31PCh. 10 - When (R)-2-butanol is left standing in aqueous...Ch. 10 - Two diastereomeric sets of enantiomers, A/B and...Ch. 10 - Acid-catalyzed dehydration of 3-methyl-2-butanol...Ch. 10 - Show how you might bring about the following...Ch. 10 - Propose a mechanism for the following pinacol...Ch. 10 - Prob. 10.37PCh. 10 - Show how each alcohol or diol can be prepared from...Ch. 10 - Dihydropyran is synthesized by treating...Ch. 10 - Show how to convert propene to each of these...Ch. 10 - Prob. 10.41PCh. 10 - Prob. 10.42PCh. 10 - The tosylate of a primary alcohol normally...Ch. 10 - Prob. 10.44PCh. 10 - Show how to convert cyclohexene to each compound...Ch. 10 - Prob. 10.46PCh. 10 - Ethanol (CH3CH2OH) and dimethyl ether (CH3OCH3)...Ch. 10 - Prob. 10.48PCh. 10 - Prob. 10.49PCh. 10 - Prob. 10.50PCh. 10 - Write the products of the following sequences of...Ch. 10 - Alcohols are important for organic synthesis,...Ch. 10 - Using your reaction roadmap as a guide, show how...Ch. 10 - Using your reaction roadmap as a guide, show how...Ch. 10 - Using your reaction roadmap as a guide, show how...Ch. 10 - Using your reaction roadmap as a guide, show how...Ch. 10 - Prob. 10.57PCh. 10 - Prob. 10.58PCh. 10 - Prob. 10.59P
Additional Science Textbook Solutions
Find more solutions based on key concepts
Q2. Which statement best defines chemistry?
a. The science that studies solvents, drugs, and insecticides
b. Th...
Introductory Chemistry (5th Edition) (Standalone Book)
45. Calculate the mass of nitrogen dissolved at room temperature in an 80.0-L home aquarium. Assume a total pre...
Chemistry: Structure and Properties
Give one example from main group chemistry that illustrates each of the following descriptions: (a) Covalent ne...
General Chemistry: Atoms First
16.43 The following pictures represent solutions at various stages in thetitration of a weak diprotic acid with...
Chemistry (7th Edition)
The chapter sections to review are shown in parentheses at the end of each problem. A "chemical-free” shampoo i...
Basic Chemistry
Draw a Lewis structure for each covalent molecule. a. HBr b. CH3F c. H2O2 d. N2H4 e. C2H6 f. CH2Cl2
Principles of General, Organic, Biological Chemistry
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Nonconjugated , -unsaturated ketones, such as 3-cyclohexenone, are in an acid-catalyzed equilibrium with their conjugated , -unsaturated isomers. Propose a mechanism for this isomerization.arrow_forwardAn unknown compound A of molecular formula C10H18O reacts with H2SO4 to form two compounds (B and C)of molecular formula C10H16. B and C both react with H2 in the presence of Pd-C to form decalin. Ozonolysis of B forms D, and ozonolysis of C forms a diketone E of molecular formula C10H16O2. Identify the structures of compounds A, B, C, and E.arrow_forwardThe reaction of 4-methylcyclohexanone with CH3MgBr followed by neutralization gives two alcohols. These two alcohols are A. enantiomers formed in equal amounts. B. diastereomers. C. constitutional isomers. D. enantiomers formed in unequal amounts.arrow_forward
- This compound can form a cyclic hemiacetal, since it has an internal OH group and an aldehyde group. What size ring would be formed from this cyclic hemiacetal? CH3CH(OH)CH2CH2CHOarrow_forwardFive isomeric alkanes (A–E) having the molecular formula C6H14 are each treated with Cl2 + hv to give alkyl halides having molecular formulaC6H13Cl. A yields five constitutional isomers. B yields four constitutionalisomers. C yields two constitutional isomers. D yields threeconstitutional isomers, two of which possess stereogenic centers. Eyields three constitutional isomers, only one of which possesses astereogenic center. Identify the structures of A–E.arrow_forwardThe shrub ma huang (Section 5.4A) contains two biologically active stereoisomers—ephedrine and pseudoephedrine—with two stereogenic centers as shown in the given structure. Ephedrine is one component of a once-popular combination drug used by body builders to increase energy and alertness, whereas pseudoephedrine is a nasal decongestant. a.Draw the structure of naturally occurring (−)-ephedrine, which has the 1R,2S configuration. b.Draw the structure of naturally occurring (+)-pseudoephedrine, which has the 1S,2S configuration. c.How are ephedrine and pseudoephedrine related? d.Draw all other stereoisomers of (−)-ephedrine and (+)pseudoephedrine, and give the R,S designation for all stereogenic centers. e.How is each compound drawn in part (d) related to (−)-ephedrine?arrow_forward
- Which of the isomeric alcohols having the molecular formula C6H14O are chiral? Which are achiral?arrow_forwardTreatment of cis-4-bromocyclohexanol with HO– affords compound A and cyclohex-3-en-1-ol. Treatment of trans-4- bromocyclohexanol under the same conditions forms compound B and cyclohex-3-en-1-ol. A and B contain different functional groups and are not isomers of each other. Propose structures for A and B and offer an explanation for their formation.arrow_forwardClopidogrel is the generic name for Plavix, a drug used to prevent the formation of blood clots in patients that have a history of heart attacks or strokes. A single enantiomer of clopidogrel can be prepared in three steps from the chiral α-hydroxy acid A. Identify B and C in the following reaction sequence, and designate the configuration of the enantiomer formed by this route as R or S.arrow_forward
- An acid–base reaction of (R)-sec-butylamine with a racemic mixture of 2-phenylpropanoic acid forms two products having different melting pointsand somewhat different solubilities. Draw the structure of these twoproducts. Assign R and S to any stereogenic centers in the products.How are the two products related? Choose from enantiomers,diastereomers, constitutional isomers, or not isomers of each other.arrow_forwardDetermine the molecular formula based on the information provided.A ketone with M+ = 192 that has4 degrees of unsaturationAn ether with M+ = 154 that has2 degrees of unsaturationarrow_forward2-Acetoxycyclohexyl tosylate reacts with acetate ion to form 1,2-cyclohexanediol diacetate. The reaction is stereospecific—that is, the stereoisomers obtained as products depend on the stereoisomer used as a reactant. Recall that because 2-acetoxycyclohexyl tosylate has two asymmetric centers, it has four stereoisomers—two are cis and two are trans. Explain the following observations:a. Both cis reactants form an optically active trans product, but each cis reactant forms a different trans product.b. Both trans reactants form the same racemic mixture.c. A trans reactant is more reactive than a cis reactant.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning

IR Spectroscopy; Author: Professor Dave Explains;https://www.youtube.com/watch?v=_TmevMf-Zgs;License: Standard YouTube License, CC-BY