
Concept explainers
Answers to all problems are at the end of this book. Detailed solutions are available in the Student Solutions Manual, Study Guide, and Problems Book.
The Structure and Ionization Properties of
Interpretation: The principal ionic species of
Concept introduction: A nucleotide refers to a basic DNA building block and structural unit. It contains a five-sided sugar, a phosphate group and a nitrogenous base. The nucleotides join together to form a chain of DNA. The nucleotides of DNA and RNA are linked covalently by bridges of the phosphate group. A phosphodiester linkage joins the
Answer to Problem 1P
The principal ionic species of
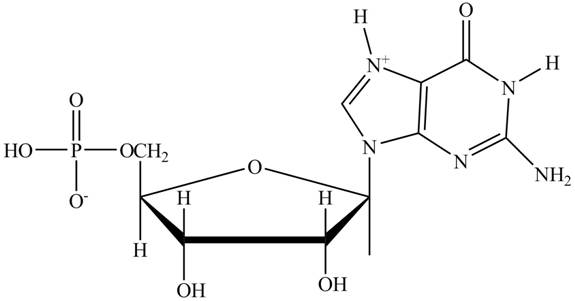
Explanation of Solution
In the polar backbone of DNA and RNA, the phosphate groups are completely ionizable and negatively charged at
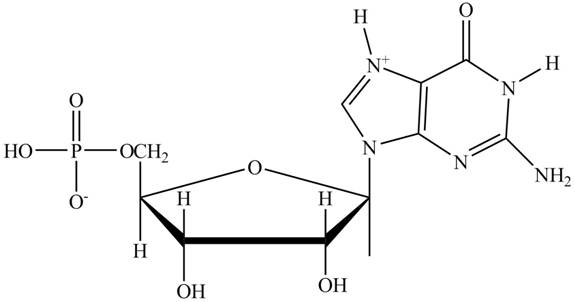
Figure 1
Want to see more full solutions like this?
Chapter 10 Solutions
Biochemistry
- Answers to all problems are at the end of this book. Detailed solutions are available in the Student Solutions Manual, Study Guide, and Problems Book. Writing Dissociation Equations for Amino Acids Write equations fur the ionic dissociations of alanine, glutamate, histidine, lysine, and phenylalanine.arrow_forwardAnswers to all problems are at the end of this book. Detailed solutions are available in the Student Solutions Manual, Study Guide, and Problems Book. Proteins and nucleic acids are informational macromolecules. What are the two minimal criteria for a linear informational polymer?arrow_forwardAnswers to all problems are at the end of this book. Detailed solutions are available in the Student Solutions Manual, Study Guide, and Problems Book. Determining the Systematic Name for a Trisaccharide Give the systematic name for it stachyose.arrow_forward
- Answers to all problems are at the end of this book. Detailed solutions are available in the Student Solutions Manual, Study Guide, and Problems Book. Oligonucleotide Structure Draw the chemical structure of pACG.arrow_forwardAnswers to all problems are at the end of this book. Detailed solutions are available in the Student Solutions Manual, Study Guide, and Problems Book. Assessing the pH Dependence of Poly-L-Glutamate Structure Poly-L glutamate adopts an tr-helical structure at low pH but becomes a random coil above pH 5. Explain this behavior.arrow_forwardAnswers to all problems are at the end of this book. Detailed solutions are available in the Student Solutions Manual, Study Guide, and Problems Book. Interpreting Kinetics Experiments from Graphical Patterns The following graphical patterns obtained from kinetic experiments have several possible interpretations depending on the nature of the experiment and the variables being plotted. Give at least two possibilities for each.arrow_forward
- Answers to all problems are at the end of this book. Detailed solutions are available in the Student Solutions Manual, Study Guide, and Problems Book. The dissociation constant for a particular protein dimer is 1 micromolar. Calculate the free energy difference for the monomer-to-dimer transition.arrow_forwardAnswers to all problems are at the end of this book. Detailed solutions are available in the Student Solutions Manual, Study Guide, and Problems Book. Calculating pH in Amino Acid Solutions II (Integrates with Chapter 2.) Calculate the pH at which the -amino group of lysine is 20% dissociated.arrow_forwardAnswers to all problems are at the end of this book. Detailed solutions are available in the Student Solutions Manual, Study Guide, and Problems Book. Draw all the possible isomers of threonine and assign (R,S) nomenclature to each.arrow_forward
- Answers to all problems are at the end of this book. Detailed solutions are available in the Student Solutions Manual, Study Guide, and Problems Book. Calculating Concentrations of Species in Amino Acid Solutions (Integrates with Chapter 2.) Calculate the concentrations of all ionic species in a 0.25 M solution of histidine at pH 2, pH 6.4, and pH 9.3.arrow_forwardAnswers to all problems are at the end of this book. Detailed solutions are available in the Student Solutions Manual, Study Guide, and Problems Book. To fully appreciate the elements of secondary structure in proteins, it is useful to have a practical sense of their structures. On a piece of paper, draw a simple but large zigzag pattern to represent a -strand. Then fill in the structure, drawing the locations of the moms of the chain on this zigzag pattern. Then draw a simple, large coil on a piece of paper to represent an -helix. Then fill in the structure, drawing the backbone atoms in the correction locations along the coil and indicating the locations of the R groups in your drawing.arrow_forwardAnswers to all problems are at the end of this book. Detailed solutions are available in the Student Solutions Manual, Study Guide, and Problems Book. The Role of Proline Residues in -Turns Pro is the amino acid least commonly found in «-helices but most commonly found in -turns. Discuss the reasons for this behavior.arrow_forward
 BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning
