Common Chemical Chemical Name Polar Non- Tonic Functional Name/ Commercia I Name Formula Covale Polar Group nt Covalen Molecu le Molecul e Chloroform CHCI, Trichloromethane T Acetone C,H,0 Propanone Hair Bleach Hydrogen Peroxide Zonrox Nacio Sodium hypochlorite Isopropyl Alcoholt or Isopropanol Ethyne or Rubbing Alcohol C,H30 Acetylene (used in welding) Acetylene Butane n-butane or (household Butane named as Botin) Formalin CH20 Formaldehyde Denatured C, H;OH Ethanol Alcohol
Common Chemical Chemical Name Polar Non- Tonic Functional Name/ Commercia I Name Formula Covale Polar Group nt Covalen Molecu le Molecul e Chloroform CHCI, Trichloromethane T Acetone C,H,0 Propanone Hair Bleach Hydrogen Peroxide Zonrox Nacio Sodium hypochlorite Isopropyl Alcoholt or Isopropanol Ethyne or Rubbing Alcohol C,H30 Acetylene (used in welding) Acetylene Butane n-butane or (household Butane named as Botin) Formalin CH20 Formaldehyde Denatured C, H;OH Ethanol Alcohol
Principles of Modern Chemistry
8th Edition
ISBN:9781305079113
Author:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Chapter14: Chemical Equilibrium
Section: Chapter Questions
Problem 93AP
Related questions
Question
Complete the following table below:
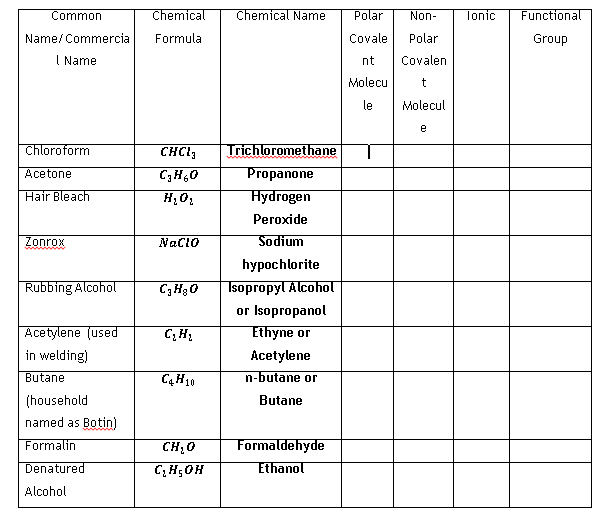
Transcribed Image Text:Common
Chemical
Chemical Name
Polar
Non-
Tonic
Functional
Name/Commercia
Formula
Covale
Polar
Group
I Name
nt
Covalen
Molecu
le
Molecul
Chloroform
CHCI,
Trichloromethane
Acetone
C;H,0
Propanone
Hair Bleach
Hydrogen
Peroxide
Zonrox
Nacio
Sodium
wwm w
hypochlorite
Isopropyl Alcohol
or Isopropanol
Ethyne or
Rubbing Alcohol
Acetylene (used
in welding)
Acetylene
Butane
C4H10
n-butane or
(household
Butane
named as Botin)
Formalin
CH 0
Formaldehyde
Denatured
Ethanol
Alcohol
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning