
Concept explainers
(a)
Interpretation:
The molecular formula of anethole has to be written.
Concept Introduction:
(a)
Explanation of Solution
The given compound is anethole. The molecular formula of anethole is
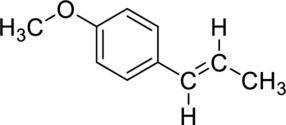
(b)
Interpretation:
The
(b)
Explanation of Solution
The functional groups present in anethole can be represented as follows,
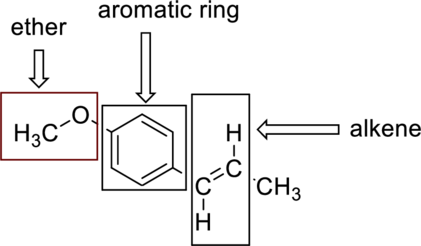
(c)
Interpretation:
The carbon-carbon double bond present in anethole has to be labeled as cis or tans.
Concept Introduction:
Cis isomer: When the two functional groups are present on the same side of the double bond, then the compound is said to be as cis isomer.
Trans isomer: When the two functional groups are present on the opposite side of the double bond, then the compound is said to be as trans isomer.
(c)
Explanation of Solution
The given compound is anethole. There is only one carbon-carbon double bond present apart from the
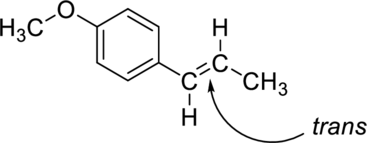
(d)
Interpretation:
The structure of anethole can be drawn and each carbon as trigonal planar of tetrahedral has to be labeled.
(d)
Explanation of Solution
The shape of
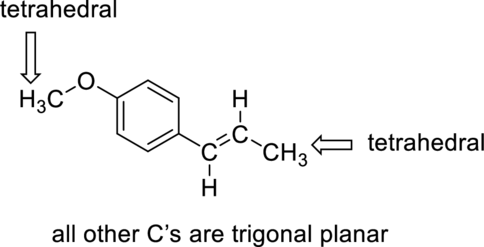
Want to see more full solutions like this?
Chapter 11 Solutions
Principles of General, Organic, Biological Chemistry
- Explain why propane boils at 42C, whereas ethanal, which has the same molecular weight, boils at 20C.arrow_forwarda.Identify the functional groups in the ball-and-stick model of elemicin, a compound partly responsible for the flavor and fragrance of nutmeg. b. Draw a skeletal structure of a constitutional isomer of elemicin that should have a higher boiling point and melting point. c. Label all electrophilic carbon atoms.arrow_forwarda. Identify the functional groups in the ball-and-stick model of elemicin, a compound partly responsible for the flavor and fragrance of nutmeg. b. Draw a skeletal structure of a constitutional isomer of elemicin that should have a higher boiling point and melting point. c. Label all electrophilic carbon atoms.arrow_forward
- The fact that sweet-tasting carbohydrates like table sugar are also high in calories has prompted the development of sweet, low-calorie alternatives. (a) Identify the functional groups in aspartame, the artificial sweetener in Equal. (b) Label all of the sites that can hydrogen bond to the oxygen atom of water. (c) Label all of the sites that can hydrogen bond to a hydrogen atom of water.arrow_forward1. What is the most dominant intermolecular forces of attraction that exist between: a. water and limonene, the major monoterpene component of lemon essential oil? b. water and constituent ions of sodium chloride? 2. Based on your answers in number 1, which pair (water-limonene or water-sodium chloride) exhibits a stronger attraction between their molecules/ions? 3. Describe what happens during the salting-out process in terms of the intermolecular forces of attraction involved as mentioned in your previous answers.arrow_forwarda.Identify the functional groups in the ball-and-stick model of neral, a compound with a lemony odor isolated from lemongrass. b. Draw a skeletal structure of a constitutional isomer of neral that should be more water soluble. c.Label the most electrophillic carbon atom.arrow_forward
- How do a carboxylic acid and an alcohol differ? Which ball-and-stick model represents a carboxylic acid and which represents an alcohol?arrow_forwardhow to draw the structures of primary, secondary and tertiary alcohol with four carbons?arrow_forwardAnalyze the structure of 4‑sec‑butyl-5-ethyl-3-methyloctane. Identify the number of ethyl and methyl groups in the compound. Note that these groups are not necessarily confined to those specifically mentioned in the name.arrow_forward
- Following is the structure of cortisone, a corticosteroid that increases the blood glucose level and stimulates the synthesis of glycogen in the liver. What functional groups are in cortisone? Group of answer choices a. ketone group, aldehyde group, and carboxyl group, b.hydroxyl group, carboxyl group, and ether group c. hydroxyl group, ketone group, and alkene group d. ester group, amide group, and alkene grouparrow_forwardWhy is ethyl alcohol soluble in water?arrow_forwardan amount of alkene requires 5.6 liters of HCl under normal conditions to fully act. The same amount, when acting with Br2, causes a weight gain of 7 grams. Find: a) the molecular formula of the alkene b) the amount of alcohol required to prepare the alkene.arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co





