
Concept explainers
Write structural formulas for each of the following:
Interpretation:
The structural formula of the given compounds is to be written.
Concept introduction:
When writing the structural formula of any compound, first the functional group from the suffix of the given name is identified.
The longest carbon chain containing the functional group is located.
The carbon atoms of the chain are numbered in a way that the functional group is at lowest numbered carbon atom.
Substituents are attached to the parent chain according to their positions given in the name.
In alkenes, the Z isomers have the higher ranked substituents on the same side of the double bond, and in E isomers, higher ranked substituents are on the opposite sides of the double bond.
Answer to Problem 26P
Solution:
a)
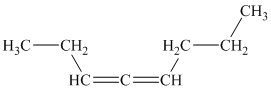
b)
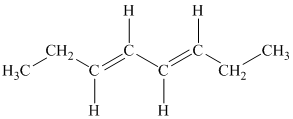
c)
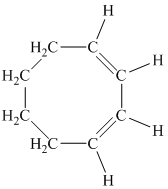
d)
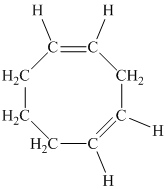
e)
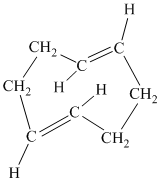
f)
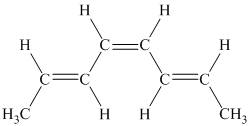
g)
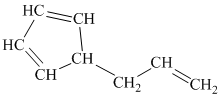
h)
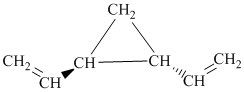
i)
Explanation of Solution
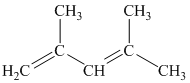
a) The given compound is
The parent name contains the word “
The structural formula is as follows:

b) The given compound is
The parent name contains the word “
The structural formula is as follows:
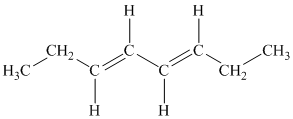
c) The given compound is
The parent name contains the word “cycloocta” in it, which means that the parent carbon chain contains eight carbon atoms in a cyclic form. The suffix added is
The structural formula is as follows:
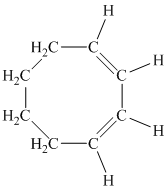
d) The given compound is
The parent name contains the word “cycloocta” in it, which means that the parent carbon chain contains eight carbon atoms in a cyclic form. The suffix added is
The structural formula is as follows:
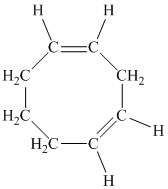
e) The given compound is
The parent name contains the word “cycloocta” in it, which means that the parent carbon chain contains eight carbon atoms in a cyclic form. The suffix added is
The structural formula is as follows:
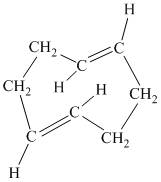
f) The given compound is
The parent name contains the word “
The structural formula is as follows:
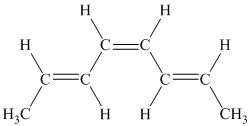
g) The given compound is
The parent name contains the word “cyclopenta” in it, which means that the parent carbon chain contains five carbon atoms in a cyclic form. The suffix added is
The structural formula is as follows:
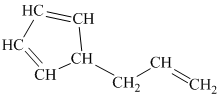
h) The given compound is
The parent name contains the word cyclopropane in it, which means that the parent carbon chain contains three carbon atoms in a cyclic form. There are two vinyl groups attached to the cyclopropane. The numbers
The structural formula is as follows:
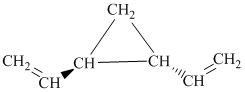
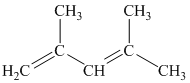
i) The given compound is
The parent name contains the word “penta” in it, which means that the longest carbon chain contains five carbon atoms. The suffix added is
The structural formula is as follows:
Want to see more full solutions like this?
Chapter 11 Solutions
Solutions Manual for Organic Chemistry
- Predict the products of the following reactions.(a) allyl bromide + cyclohexyl magnesium bromide (b) cyclopentadiene + anhydrous HCl(c) 2@methylpropene + NBS, light (d) furan + trans@1,2@dicyanoethylene(e) buta@1,3@diene + bromine water (f) hexa@1,3,5@triene + bromine in CCl4arrow_forwardThe following ultraviolet absorption maxima have been measured: 1,3-Butadiene 217 nm 2-Methyl-1,3-butadiene 220 nm 1,3-Pentadiene 223 nm 2,3-Dimethyl-1,3-butadiene 226 nm 2,4-Hexadiene 227 nm 2,4-Dimethyl-1,3-pentadiene 232 nm 2,5-Dimethyl-2,4-hexadiene 240 nm What conclusion can you draw about the effect of alkyl substitution on UV absorption maxima? Approximately what effect does each added alkyl group have?arrow_forwardUsing cyclooctyne as your starting material, show how you would synthesize the following compounds. (Once you haveshown how to synthesize a compound, you may use it as the starting material in any later parts of this problem.)(a) cis-cyclooctene (b) cyclooctane (c) trans-1,2-dibromocyclooctane(d) cyclooctanone (e) 1,1-dibromocyclooctane (f) 3-bromocyclooctene(g) cyclooctane-1,2-dionearrow_forward
- 5 Predict the products of the following reactions. buta@1,3@diene + bromine waterarrow_forwardClassify the following dienes and polyenes as isolated, conjugated, cumulated, or some combination of theseclassifications.(a) cycloocta-1,4-diene (b) cycloocta-1,3-diene (c) cyclodeca-1,2-diene(d) cycloocta-1,3,5,7-tetraenearrow_forward1)Chemistry students are taking an experimental course in organic chemistry at a public university. During an experiment involving conjugated dienes, some doubts arose when discussing the results obtained so far: (a) A student obtained two products from the reaction of 1,3-cyclohexadiene with Br2. His lab partner was surprised to get only one product from the reaction of 1,3 - cyclohexadiene with HBr. Explain these distinct results. (b) One student, seeing the discussion of colleagues, commented that she obtained two distinct products when reacting 1,3,5-hexatriene with HBr, with different yields just by changing the reaction temperature. Explain the results she obtained using reaction mechanism and based on kinetic and thermodynamic control involving conjugated dienes.arrow_forward
- TRUE OR FALSE i. A =C-Br bond is more polar than a -C-Br bond. ii. A bromonium ion can undergo hydride or methyl shift to produce a more stable carbocation. iii. The two conformers of cis-1,3-diaminocyclohexane will have equal stability.arrow_forward(1) Predict the outcome of the addition of HBr to (a) trans-2-pentene, (b) 2-methyl-2-butene, and (c) 4-methylcyclohexene. How many isomers can be formed in each case? (2) Addition of HBr to 3,3-dimethyl-1-butene gives a mixture of two isomeric alkyl bromide products. Draw structures for the two products, and give a mechanistic explanation for their formation.arrow_forwardWrite structural formulas for the products that form when 1-butene reacts with each of the following reagents: (a) HI (b) H2, Pt (c) Dilute H2SO4, warm (d) Cold concentrated H2SO4 (e) Cold concentrated H2SO4, then H2O and heat (f) HBr (g) Br2 in CCl4 (h) Br2 in H2O (i) HCl (j) O3, then Me2S (k) OsO4, then NaHSO3/H2O (l) KMnO4, OH-, heat, then H3O+ (m) Hg(OAc)2 in THF and H2O, then NaBH4, OH- (n) BH3:THF, then H2O2, OH-arrow_forward
- 1. Consider the SN2 reaction between the two molecules shown: ( in picture) a) Find nucleophile in reaction.b) find leaving group in the reaction.c) Draw the curved arrows indicating the bonds broken and formed in the reaction to correctly show how this mechanism proceeds. d) Draw only the structures of the product(s), including all non-zero formal charges and relevantlone pairs.arrow_forwardPredict the products of the following reactions. cyclopentadiene + methyl acrylate, CH2“CH¬COOCH3arrow_forwardFirst Writedown which reaction it is? SN1, SN2, E1? Write a stepwise mechanism for the following reactions showing ALL intermediates. Use curved arrows to symbolize the flow of electrons to show how each of the intermediates and product are formed. Show all necessary lone pairs and formal charges.arrow_forward
