
Concept explainers
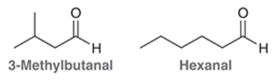
When a consumer purchases a tomato, smell is one of the factors affecting selection. Researchers in Japan have thus analyzed the volatile components of tomatoes using a novel method.11 Among the complex mixture of 367 compounds detected were 3-methylbutanal and hexanal. Propose a single synthesis that produces an equimolar mixture of these two compounds (equal amounts of both products) starting with one
Want to see the full answer?
Check out a sample textbook solution
Chapter 11 Solutions
ORGANIC CHEMISTRY (LL)-W/WILEYPLUS
- Following is the structural formula of the tranquilizer meparfynol (Oblivon). Propose a synthesis for this compound starting with acetylene and a ketone. (Notice the -yn- and -ol in the chemical name of this compound, indicating that it contains alkyne and hydroxyl functional groups.)arrow_forwardWhich of the following reactions will synthesize phenol from benzene? 1) HNO3 + H2SO4; 2) Fe, HCl; 3) NaNO2, HCl, 0-10 oC; 4) warm H2SO4 and H2O 1) HNO3 + H2SO4; 2) Fe, HCl; 3) NaNO2, HCl, 0-10 oC; 4) CuCN; 5) dilute acid and heat 1) Acetyl chloride & AlCl3; 2) bleach 1) Ph-N2+ + KI; 2) BrMgCH=CH2 in ether, followed by H3O+; 3) warm, conc'd KMnO4 1) Cl-CH(CH3)-CH2CH2CH3 + FeBr3; 2) hot, conc'd KMnO4arrow_forwardFor this question, n-Butanol (CH3CH2CH2CH2OH) and t-butanol ((CH3)3COH) are converted to their corresponding chlorides on being reacted with hydrogen chloride (HCl). Please answer the following Write out an equation for each reaction Assign each the appropriate symbol (SN1 or SN2) Write a suitable mechanism for each reactionarrow_forward
- A student attempted to prepare 1-chlorobutane by mixing 1-butanol with NaCl in acetone. Was the student succesful? Explain.arrow_forwardWhich of the following statements is true10. As the alkyl chain increases in molecular weight among primary alcohol, the solubility in water increases. 11. An ethoxide ion exhibits greater proton-accepting property compared to an ethyloxonium ion. 12. Phenol is more acidic than cyclohexyl alcohol due to the electron-donating ability of phenolic oxygen.arrow_forwardPropose an efficient synthesis for two of the following target molecules, starting with any of the following starting materials for the source of carbon atoms: (i) benzene, (i) any alcohol containing four or fewer carbon atoms, (im) carbon dioxide, (iv) cyanide ion. This means that any carbon atom that ends up in the target molecule must come from one of these starting materials! Br (а) H2N (b) HOarrow_forward
- Draw a diagram similar to that in figure 6 detailing the separation of 1,2-dichlorobenzene, phenol (pka = 10), and pyridine by an extraction protocol. OH aa CI 1,2-dichlorobenzene phenol ·N·. pyridinearrow_forwardPredict the major organic product of the reaction sequence described. Styrene (1.53 g, 0.011 mol) in methanol (30 mL) was added to a mixture of Hg(OAc)2 (5.30 g, 0.016 mol) in methanol (100 mL) at room temp. and stirred for 24 hours. Sodium hydroxide (3.0 M, 16 mL) was added, followed by NaBH4 (0.32 g, 0.008 mol) in NaOH (3.0 M, 16 mL) at 0 °C. The precipitated Hg was removed by filtration. The product was isolated by diethyl ether extraction. After drying over Na2SO4, solvent was removed and distillation gave the productarrow_forwardUsing your reaction roadmap as a guide, show how to convert ethylene into 1-butene. All of the carbon atoms of the target molecule must be derived from ethylene. Show all intermediate molecules synthesized along the way. Ethylene 1-Butenearrow_forward
- Bb.55.arrow_forward›Predict the major organic product of the reaction sequence described. Styrene (1.53 g, 0.011 mol) in methanol (30 mL) was added to a mixture of Hg(OAc)₂(5.30 g, 0.016 mol) in methanol (100 mL) at room temp. and stirred for 24 hours. Sodium hydroxide (3.0 M, 16 mL) was added, followed by NaBH (0.32 g, 0.008 mol) in NaOH (3.0 M, 16 mL) at 0 °C. The precipitated Hg was removed by filtration. The product was isolated by diethyl ether extraction. After drying over Na₂ SO4, solvent was removed and distillation gave the product. (Adapted from: Senda, Y.; Kanto, H.; Itoh, H. J. Chem. Soc., Perkin Trans. 2 1997, 1143–1146.) Draw the major organic product. H₂O Harrow_forwardAlcohols are important for organic synthesis, especially in situations involving alkenes. The alcohol might be the desired product, or the OH group might be transformed into another functional group via halogenation, oxidation, or perhaps conversion to a sulfonic ester derivative. Formation of an alcohol from an alkene is particularly powerful because conditions can be chosen to produce either the Markovnikov or non-Markovnikov product from an unsymmetrical alkene. Using your reaction roadmap as a guide, show how to convert 4-methyl-1-pentene into 5-methylhexanenitrile. You must use 4-methyl-1-pentene and sodium cyanide as the source of all carbon atoms in the target molecule. Show all reagents needed and all molecules synthesized along the way.arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

