
a)
Interpretation: The dissolution of the following in water or
Concept Introduction:
The bonds involving the atoms with various electronegativites having a large dipole moment are called as polar solvents. Example of polar solvent is Water.
The bonds involving the atoms with same electronegativites are called as non polar solvents. Examples of non polar solvents are Gasoline, Carbon tetrachloride etc
Water is a polar solvent as well as dissolves polar solutes and ionic solutes. Carbon tetrachloride
(
a)
Answer to Problem 51E
Answer
Explanation of Solution
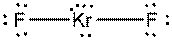
b)
Interpretation: The dissolution of the following in water or
Concept Introduction:
The bonds involving the atoms with various electronegativites having a large dipole moment are called as polar solvents. Example of polar solvent is Water.
The bonds involving the atoms with same electronegativites are called as non polar solvents. Examples of non polar solvents are Gasoline, Carbon tetrachloride etc
Water is a polar solvent as well as dissolves polar solutes and ionic solutes. Carbon tetrachloride
(
b)
Answer to Problem 51E
Answer
Explanation of Solution
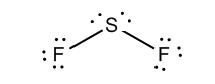
c)
Interpretation: The dissolution of the following in water or
Concept Introduction:
The bonds involving the atoms with various electronegativites having a large dipole moment are called as polar solvents. Example of polar solvent is Water.
The bonds involving the atoms with same electronegativites are called as non polar solvents. Examples of non polar solvents are Gasoline, Carbon tetrachloride etc
Water is a polar solvent as well as dissolves polar solutes and ionic solutes. Carbon tetrachloride
(
c)
Answer to Problem 51E
Answer
Explanation of Solution
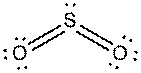
d).
Interpretation: The dissolution of the following in water or
Concept Introduction:
The bonds involving the atoms with various electronegativites having a large dipole moment are called as polar solvents. Example of polar solvent is Water.
The bonds involving the atoms with same electronegativites are called as non polar solvents. Examples of non polar solvents are Gasoline, Carbon tetrachloride etc
Water is a polar solvent as well as dissolves polar solutes and ionic solutes. Carbon tetrachloride
(
d).
Answer to Problem 51E
Answer
Explanation of Solution
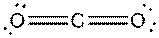
e)
Interpretation: The dissolution of the following in water or
Concept Introduction:
The bonds involving the atoms with various electronegativites having a large dipole moment are called as polar solvents. Example of polar solvent is Water.
The bonds involving the atoms with same electronegativites are called as non polar solvents. Examples of non polar solvents are Gasoline, Carbon tetrachloride etc
Water is a polar solvent as well as dissolves polar solutes and ionic solutes. Carbon tetrachloride
(
e)
Answer to Problem 51E
Answer
Explanation of Solution
f).
Interpretation: The dissolution of the following in water or
Concept Introduction:
The bonds involving the atoms with various electronegativites having a large dipole moment are called as polar solvents. Example of polar solvent is Water.
The bonds involving the atoms with same electronegativites are called as non polar solvents. Examples of non polar solvents are Gasoline, Carbon tetrachloride etc
Water is a polar solvent as well as dissolves polar solutes and ionic solutes. Carbon tetrachloride
(
f).
Answer to Problem 51E
Answer
Explanation of Solution
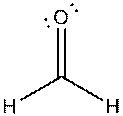
g)
Interpretation: The dissolution of the following in water or
Concept Introduction:
The bonds involving the atoms with various electronegativites having a large dipole moment are called as polar solvents. Example of polar solvent is Water.
The bonds involving the atoms with same electronegativites are called as non polar solvents. Examples of non polar solvents are Gasoline, Carbon tetrachloride etc
Water is a polar solvent as well as dissolves polar solutes and ionic solutes. Carbon tetrachloride
(
g)
Answer to Problem 51E
Answer
Explanation of Solution
Want to see more full solutions like this?
Chapter 11 Solutions
Chemistry
- You dissolve 1.0 mol of urea (H2NCONH2) in 270 g of water. The mole fraction of urea is (a) 3710 3 (b) 0.063 (c) 16 (d) 1.0 and its molality is (a) 1.0 (b) 3.7 (c) 0.063 (d) 0.27arrow_forwardStarch contains CC, CH, CO, and OH bonds. Hydrocarbons have only CC and CH bonds. Both starch and hydrocarbons can form colloidal dispersions in water. Which dispersion is classified as hydrophobic? Which is hydrophilic? Explain briefly.arrow_forwardDistinguish between dispersion methods and condensation methods for preparing colloidal systems.arrow_forward
- Equal numbers of moles of two soluble, substances, substance A and substance B, are placed into separate 1.0-L samples of water. a The water samples are cooled. Sample A freezes at 0.50C, and Sample B freezes at l.00C. Explain how the solutions can have different freezing points. b You pour 500 mL of the solution containing substance B into a different beaker. How would the freezing point of this 500-mL portion of solution B compare to the freezing point of the 1.0-L sample of solution A? c Calculate the molality of the solutions of A and B. Assume that i = 1 for substance A. d If you were to add an additional 1.0 kg of water to solution B, what would be the new freezing point of the solution? Try to write an answer to this question without using a mathematical formula. e What concentration (molality) of substances A and B would result in both solutions having a freezing point of 0.25C? f Compare the boiling points, vapor pressure, and osmotic pressure of the original solutions of A and B. Dont perform the calculations; just state which is the greater in each ease.arrow_forwardA compound of cobalt, carbon, and oxygen contains 28.10% C and 34.47% Co. When 0.147 g of this compound is dissolved in 6.72 g of cyclohexane, the solution freezes at 5.23C. What is the molecular formula of this compound?arrow_forwardAn organic compound contains carbon (71.17%), hydrogen (5.12%) with the remainder nitrogen. Dissolving 0.177 g of the compound in 10.0 g of benzene gives a solution with a vapor pressure of 94.16 mm Hg at 25 C. (The vapor pressure of pure benzene at this temperature is 95.26 mm Hg.) What is the molecular formula for the compound?arrow_forward
- Calculate the enthalpies of solution for Li2SO4 and K2SO4. Are the solution processes exothermic or endothermic? Compare them with LiCl and KCl. What similarities or differences do you find?arrow_forwardA water solution of sodium hypochlorite (NaOCl) is used as laundry bleach. The concentration of sodium hypochlorite is 0.75 m. Express this concentration as a mole fraction.arrow_forwardHow much N2 can dissolve in water at 25 C if the N2 partial pressure is 585 mm Hg?arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
 Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co





