
Without referring to your textbook or a periodic table, write the full electron configuration, the orbital box diagram, and the noble gas shorthand configuration for the elements with the following
msp;
(a)
Interpretation:
The electronic configuration of the given element, the orbital box diagram and the noble gas shorthand configuration for the elements are to be stated.
Concept Introduction:
The distribution of the electrons that exists in the atomic orbital of an atom is collectively known as electronic configuration. The description of every electron in an orbital is given by the electronic configuration of that atom.
Answer to Problem 97AP
The electronic configuration of the given element with
Explanation of Solution
The electronic configuration of the given element with
The orbitals in the orbital box diagram are arranged in increasing order of energy shells. The orbital box diagram is shown in figure 1.
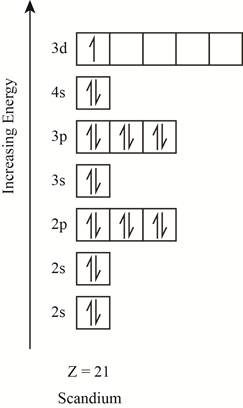
Figure 1
The electronic configuration of the given element can also be written as
(b)
Interpretation:
The electronic configuration of the given element, the orbital box diagram and the noble gas shorthand configuration for the elements are to be stated.
Concept Introduction:
The distribution of the electrons that exists in the atomic orbital of an atom is collectively known as electronic configuration. The description of every electron in an orbital is given by the electronic configuration of that atom.
Answer to Problem 97AP
The electronic configuration of the given element with
Explanation of Solution
The electronic configuration of the given element with
The orbitals in the orbital box diagram are arranged in increasing order of energy shells. The orbital box diagram is shown in figure 2.
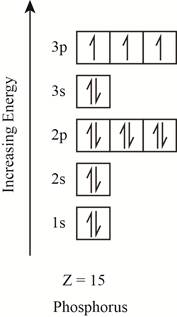
Figure 2
The electronic configuration of the given element can also be written as
(c)
Interpretation:
The electronic configuration of the given element, the orbital box diagram and the noble gas shorthand configuration for the elements are to be stated.
Concept Introduction:
The distribution of the electrons that exists in the atomic orbital of an atom is collectively known as electronic configuration. The description of every electron in an orbital is given by the electronic configuration of that atom.
Answer to Problem 97AP
The electronic configuration of the given element with
Explanation of Solution
The electronic configuration of the given element with
The orbitals in the orbital box diagram are arranged in increasing order of energy shells. The orbital box diagram is shown in figure 3.
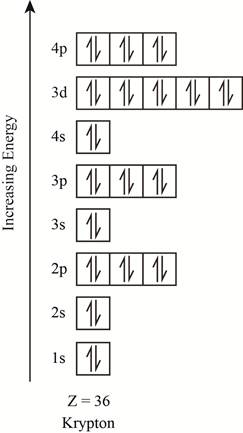
Figure 3
The electronic configuration of the given element can also be written as
(d)
Interpretation:
The electronic configuration of the given element, the orbital box diagram and the noble gas shorthand configuration for the elements are to be stated.
Concept Introduction:
The distribution of the electrons that exists in the atomic orbital of an atom is collectively known as electronic configuration. The description of every electron in an orbital is given by the electronic configuration of that atom.
Answer to Problem 97AP
The electronic configuration of the given element with
Explanation of Solution
The electronic configuration of the given element with
The orbitals in the orbital box diagram are arranged in increasing order of energy shells. The orbital box diagram is shown in figure 4.
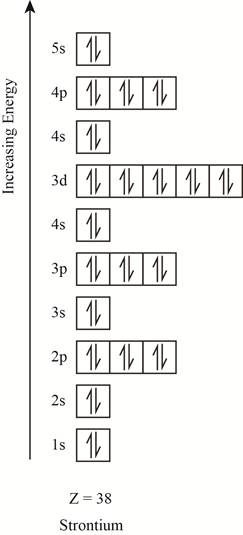
Figure 4
The electronic configuration of the given element can also be written as
(e)
Interpretation:
The electronic configuration of the given element, the orbital box diagram and the noble gas shorthand configuration for the elements are to be stated.
Concept Introduction:
The distribution of the electrons that exists in the atomic orbital of an atom is collectively known as electronic configuration. The description of every electron in an orbital is given by the electronic configuration of that atom.
Answer to Problem 97AP
The electronic configuration of the given element with
Explanation of Solution
The electronic configuration of the given element with
The orbitals in the orbital box diagram are arranged in increasing order of energy shells. The orbital box diagram is shown in figure 5.
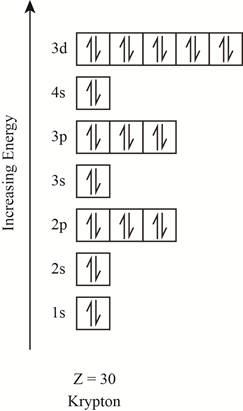
Figure 5
The electronic configuration of the given element can also be written as
Want to see more full solutions like this?
Chapter 11 Solutions
Introductory Chemistry: A Foundation
- List the orbitals in order of increasing orbital energy up to and including 3p orbitals.arrow_forwardGive electron configurations according to the Bohr model for each of the following elements. Try to not use Figure 3.11, but instead determine the configuration based on your knowledge of the number of electrons in each atom and the maximum number of electrons in each Bohr orbit. Indicate which of these elements you expect to be the most reactive and the least reactive. a. B b. Si c. Ca d. F e. Ararrow_forwardWhich of the following statements is(are) true? a. The 2s orbital in the hydrogen atom is larger than the 3s orbital also in the hydrogen atom. b. The Bohr model of the hydrogen atom has been found to be incorrect. c. The hydrogen atom has quantized energy levels. d. An orbital is the same as a Bohr orbit. e. The third energy level has three sublevels, the s, p, and d sublevels.arrow_forward
- Using only the periodic table inside the front cover of the text, write the expected ground-state electron configurations for a. the third element in Group 5A. b. element number 116. c. an element with three unpaired 5d electrons. d. the halogen with electrons in the 6p atomic orbitals.arrow_forwardWhen describing the electrons in an orbital, we use arrows pointing upward and downward ( and ) to indicate what property?arrow_forwardWhich orbital is the first be filled in any atom? Why?arrow_forward
 World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
 Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning





