
A commercial refrigerator with refrigerant-134a as the working fluid is used to keep the refrigerated space at −30°C by rejecting its waste heat to cooling water that enters the condenser at 18°C at a rate of 0.25 kg/s and leaves at 26°C. The refrigerant enters the condenser at 1.2 MPa and 65°C and leaves at 42°C. The inlet state of the compressor is 60 kPa and −34°C and the compressor is estimated to gain a net heat of 450 W from the surroundings. Determine (a) the quality of the refrigerant at the evaporator inlet, (b) the refrigeration load, (c) the COP of the refrigerator, and (d) the theoretical maximum refrigeration load for the same power input to the compressor.
FIGURE P11–22
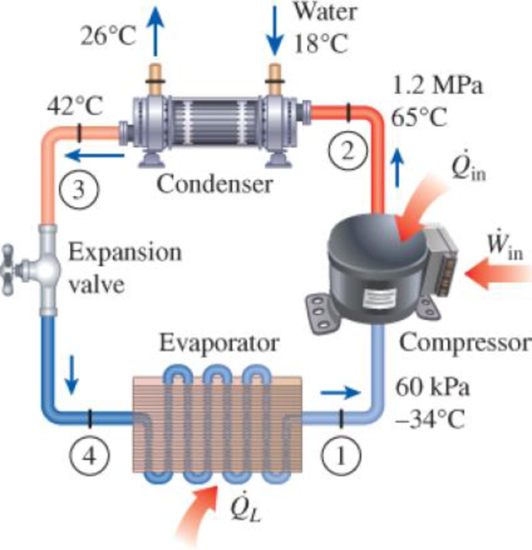
(a)
The quality of the refrigerant at the evaporator inlet.
Answer to Problem 22P
The quality of the refrigerant at the evaporator inlet is
Explanation of Solution
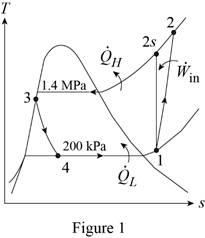
Show the T-s diagram for the refrigeration cycle as in Figure (1).
Express specific enthalpy at state 3.
Here, specific enthalpy at saturated liquid and temperature of
Express the quality of the refrigerant at the evaporator inlet.
Here, specific enthalpy at saturated liquid and pressure of
Conclusion:
Perform unit conversion of pressure at state 1 from
Refer Table A-13, “superheated refrigerant-134a”, and write the value of specific enthalpy at state 1
Write the formula of interpolation method of two variables.
Here, the variables denote by x and y is initial temperature and specific enthalpy at state 1 respectively.
Show the specific enthalpy at state 2 corresponding to specific entropy as in Table (1).
|
Initial temperature |
Specific enthalpy at state 1 |
| 227.80 | |
| 240.78 |
Substitute
Perform unit conversion of pressure at state 2 from
Refer Table A-13, “superheated refrigerant-134a”, and write the value of specific enthalpy at state 2
Show the specific enthalpy at state 2 corresponding to temperature as in Table (2).
|
Temperature |
Specific enthalpy at state 2 |
| 289.66 | |
| 300.63 |
Use Excel by taking the values from Table (2), and using Equation (III) to get specific enthalpy at state 2.
Refer Table A-11, “saturated refrigerant 134a-temperature table”, and write the properties corresponding to temperature at state 3 of
Substitute
From Figure (1), write the specific enthalpy at state 3 is equal to state 4 due to throttling process.
Here, specific enthalpy at state 4 is
Refer Table A-12, “saturated refrigerant 134a-pressure table”, and write the properties corresponding to pressure at state 4 of
Substitute
Hence, the quality of the refrigerant at the evaporator inlet is
(b)
The refrigeration load.
Answer to Problem 22P
The refrigeration load is
Explanation of Solution
Express the mass flow rate of the refrigerant from an energy balance on the compressor.
Here, mass flow rate of the water is
Express the rate of heat supplied from the refrigerant.
Express compressor power input.
Here, rate of heat gained by compressor is
Express the refrigeration load.
Conclusion:
Refer Table A-4, “saturated water-temperature table”, and write the initial specific enthalpy of water corresponding to temperature of
Show the initial specific enthalpy of water corresponding to temperature as in Table (3).
|
Temperature |
Initial specific enthalpy of water |
| 15 | 62.982 |
| 18 | |
| 20 | 83.915 |
Use Excel by taking the values from Table (3), and using Equation (III) to get initial specific enthalpy of water.
Refer Table A-4, “saturated water-temperature table”, and write the initial specific enthalpy of water corresponding to temperature of
Show the initial specific enthalpy of water corresponding to temperature as in Table (3).
|
Temperature |
Final specific enthalpy of water |
| 25 | 104.83 |
| 26 | |
| 30 | 125.74 |
Use Excel by taking the values from Table (3), and using Equation (III) to get final specific enthalpy of water.
Substitute
Substitute
Substitute
Substitute
Hence, the refrigeration load is
(c)
The COP of the refrigerator.
Answer to Problem 22P
The COP of the refrigerator is
Explanation of Solution
Express the coefficient of performance of the refrigerator.
Conclusion:
Substitute
Hence, the coefficient of performance of the refrigerator is
(d)
The theoretical maximum refrigeration load.
Answer to Problem 22P
The theoretical maximum refrigeration load is
Explanation of Solution
Express the reversible COP of the refrigerator for the similar temperature limits.
Here, high and low source temperature is
Express the theoretical maximum refrigeration load.
Conclusion:
Substitute
Substitute
Hence, the theoretical maximum refrigeration load is
Want to see more full solutions like this?
Chapter 11 Solutions
Thermodynamics: An Engineering Approach
- A commercial refrigerator with refrigerant-134a as the working fluid is used to keep the refrigerated space at –35°C by rejecting waste heat to cooling water that enters the condenser at 18°C at a rate of 0.25 kg/s and leaves at 26°C. The refrigerant enters the condenser at 1.2 MPa and 50°C and leaves at the same pressure subcooled by 5°C. If the compressor consumes 3.3 kW of power, determine the minimum power input to the compressor for the same refrigeration load.arrow_forwardConsider a refrigrator that operates on the vapor compression refrigeration cycle with R-134a as the working fluid. The refrigerant enters the compressor as saturated vapor at 70 kPa, and exits at 1200 kPa and 90°C, and leaves the condenser as saturated liquid at 1200 kPa. The coefficient of performance of this refrigrator isarrow_forwardRefrigerant-134a enters the compressor of a refrigerator at 0.14MPa and -10°C at a rate of 0.05 kg/s and leaves at 0.8 MPa and 50°C. The refrigerant is cooled in the condenser to 26°C and 0.72 MPa and is throttled to 0.15 MPa. Determine (a) the rate of heat removal from the refrigerated space and the power input to the compressor, (b) the isentropic efficiency of the compressor, and (c) the coefficient of performance of the refrigerator. Answers: (a) 7.93 kW, 2.02 kW, (b) 0.939, (c) 3.93arrow_forward
- A commercial refrigerator with refrigerant-134a as the working fluid is used to keep the refrigerated space at –30°C by rejecting its waste heat to cooling water that enters the condenser at 18°C at a rate of 0.25 kg/s and leaves at 26°C. The refrigerant enters the condenser at 1.2 MPa and 65°C and leaves at 42°C. The inlet state of the compressor is 60 kPa and –34°C and the compressor is estimated to gain a net heat of 450 W from the surroundings. Determine the theoretical maximum refrigeration load for the same power input to the compressor. Water 26°C ↑ 18°C 1.2 MPа 42°C 65°C Condenser Expansion Win valve Evaporator Compressor 60 kPa -34°Carrow_forwardAn air conditioner using refrigerant-134a as the working fluid is used to keep the temperature of a room at 23°C by giving heat to the external environment at 37°C. The heat gain of the house from the walls and windows is 250 kJ/min; 900 W heat is emitted into the room from the computer, TV and lamps. The refrigerant enters the compressor with a flow rate of 100 L/min in the form of saturated vapor at 400 kPa pressure and leaves the compressor at 70°C at 1200 kPa pressure.a) Draw the cycle by showing the elements of the cycle.b) the actual COP value,c) The highest COP value,d) The smallest refrigerant can have for the same compressor inlet and outlet conditions.Calculate the volumetric flow.arrow_forwardConsider a refrigerator that operates on the vapor compression refrigeration cycle with R-134a as the working fluid. The refrigerant enters the compressor as a saturated vapor at 140 kPa, and exits at 800 kPa and 60 degrees Celsius, and leaves the condenser as a saturated liquid at 800 kPa. The COP of this refrigerator is:arrow_forward
- A commercial refrigerator with refrigerant-134a as the working fluid is used to keep the refrigerated space at -35°C by rejecting waste heat to cooling water that enters the condenser at 18°C at a rate of 0.25 kg/s and leaves at 26°C. The refrigerant enters the condenser at 1.2 MPa and 50°C and leaves at the same pressure subcooled by 5°C. If the compressor consumes 3.3 kW of power, determine (a) the mass flow rate of the refrigerant, (b) the refrigeration load, (c) the COP, and (d) the minimum power input to the compressor for the same refrigeration load.arrow_forwardA refrigerated room is kept at −18◦C by a vapor-compression cycle with R-134a as the refrigerant. Heat is rejected to cooling water that enters the condenser at 14◦C at a rate of 0.35 kg/s and leaves at 22◦C. The refrigerant enters the condenser at 1.2MPa and 50◦C and leaves at the same pressure subcooled by 5◦C. If the compressor consumes 5.5 kW of power, determine (a) the mass flow rate of the refrigerant, (b) the refrigeration load and the COP, (c) the second-law efficiency of the refrigerator and the total exergy destruction in the cycle, and (d) the exergy destruction in the condenser. Take specific heat of water to be 4.18 kJ/kg·◦C.arrow_forwardRefrigerant-134a enters the compressor of a refrigerator at 140 kPa and -10°C at a rate of 0.3 m3/min and leaves at 1 MPa. The isentropic efficiency of the compressor is 78 percent. The refrigerant enters the throttling valve at 0.95 MPa and 30°C and leaves the evaporator as saturated vapor at -18.5°C. Show the cycle on a T-s diagram with respect to saturation lines, and determine (a) the power input to the compressor, (b) the rate of heat removal from the refrigerated space, and (c) the pressure drop andrate of heat gain in the line between the evaporator and the compressor. answers 1.88 kW, 7.11 kW, 1.72 kPa, 0.24 kWarrow_forward
- A refrigerator uses R-134a as the working fluid and operates on an ideal vapor compression cycle between 0.14 MPa and 0.8 MPa. If the mass flow rate of the refrigerant is 0.06 kg/s, determine (a) the rate of heat removal from the refrigerated space, (b) the power input to the compressor, (c) the heat rejection rate in the condenser, and (d) the COP.arrow_forwardA Carnot refrigeration cycle is executed in a closed system in the saturated liquid-vapor mixture region using 1 kg of refrigerant-134a as the working fluid. The maximum and the minimum temperatures in the cycle are 22C and -4C, respectively. If the refrigerant is saturated liquid at the end of the heat rejection process, and the net work input to the cycle is 16 kJ, determine: b) The pressure at the end of the heat rejection process. Should correct please. (Gpt/ai Answer not allowed)arrow_forwardA commercial refrigerator using R-134a as a refrigerant is used to keep the cooled environment at -35 C. The refrigerator releases the waste heat to the cooling water that enters the condenser at 18 C at 0.25 kg / s and leaves the condenser at 26 C. The refrigerant enters the condenser at a pressure of 1.2 MPa and a temperature of 50 ° C, and leaves the condenser at the same pressure, supercooled at 5 ° C. Since the compressor consumes 3.3 kW of power, a) Mass flow rate of the refrigerant, b) Cooling load and COP value c) Calculate the minimum power consumed by the compressor for the same cooling load.arrow_forward
 Elements Of ElectromagneticsMechanical EngineeringISBN:9780190698614Author:Sadiku, Matthew N. O.Publisher:Oxford University Press
Elements Of ElectromagneticsMechanical EngineeringISBN:9780190698614Author:Sadiku, Matthew N. O.Publisher:Oxford University Press Mechanics of Materials (10th Edition)Mechanical EngineeringISBN:9780134319650Author:Russell C. HibbelerPublisher:PEARSON
Mechanics of Materials (10th Edition)Mechanical EngineeringISBN:9780134319650Author:Russell C. HibbelerPublisher:PEARSON Thermodynamics: An Engineering ApproachMechanical EngineeringISBN:9781259822674Author:Yunus A. Cengel Dr., Michael A. BolesPublisher:McGraw-Hill Education
Thermodynamics: An Engineering ApproachMechanical EngineeringISBN:9781259822674Author:Yunus A. Cengel Dr., Michael A. BolesPublisher:McGraw-Hill Education Control Systems EngineeringMechanical EngineeringISBN:9781118170519Author:Norman S. NisePublisher:WILEY
Control Systems EngineeringMechanical EngineeringISBN:9781118170519Author:Norman S. NisePublisher:WILEY Mechanics of Materials (MindTap Course List)Mechanical EngineeringISBN:9781337093347Author:Barry J. Goodno, James M. GerePublisher:Cengage Learning
Mechanics of Materials (MindTap Course List)Mechanical EngineeringISBN:9781337093347Author:Barry J. Goodno, James M. GerePublisher:Cengage Learning Engineering Mechanics: StaticsMechanical EngineeringISBN:9781118807330Author:James L. Meriam, L. G. Kraige, J. N. BoltonPublisher:WILEY
Engineering Mechanics: StaticsMechanical EngineeringISBN:9781118807330Author:James L. Meriam, L. G. Kraige, J. N. BoltonPublisher:WILEY





