
Organic Chemistry
8th Edition
ISBN: 9781305580350
Author: William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Chapter 11.9, Problem AQ
Interpretation Introduction
Interpretation:
The stereochemical outcome of the reaction at boxed and circled carbons relative to the reactant has to be discussed.
Concept Introduction:
Stereochemistry: The spatial arrangement of atoms or groups present in compound.
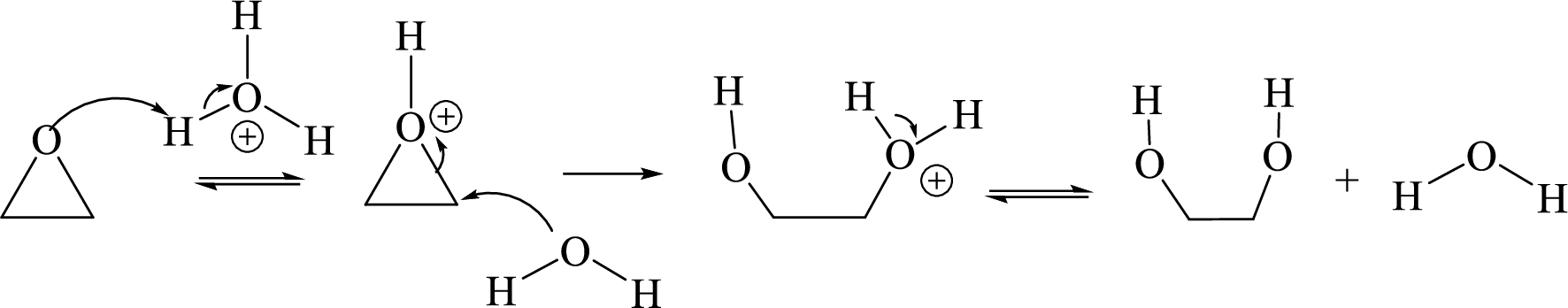
Ring opening: Ring opening of epoxide takes place by acid-catalyzed hydrolysis of an epoxide.
Acid-catalyzed hydrolysis of an epoxide:
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
Draw the major product (with stereochemistry) of the following reaction. Draw only one enantiomer for a racemic mixture.
Which reactions are stereoselective?
When a carbonyl compound is attacked by a nucleophile resulting in the formation of a new stereogenic center, what product(s) will be formed?
A) A racemic mixture of the two possible enantiomers
B) Contains more of one enantiomer than another because of steric reasons around the carbonyl
C) Contains more of one enantiomer than another depending on the temperature of the reaction
D) Contains different products depending on the solvent used
Chapter 11 Solutions
Organic Chemistry
Ch. 11.2 - Write IUPAC and common names for these ethers. (a)...Ch. 11.3 - Arrange these compounds in order of increasing...Ch. 11.4 - Show how you might use the Williamson ether...Ch. 11.4 - Show how ethyl hexyl ether might be prepared by a...Ch. 11.5 - Account for the fact that treatment of tert-butyl...Ch. 11.5 - Draw structural formulas for the major products of...Ch. 11.6 - Prob. 11.7PCh. 11.8 - Draw the expected products of Sharpless...Ch. 11.9 - Prob. AQCh. 11.9 - Prob. BQ
Ch. 11.9 - Prob. CQCh. 11.9 - Prob. DQCh. 11 - Write names for these compounds. Where possible,...Ch. 11 - Prob. 11.11PCh. 11 - Each compound given in this problem is a common...Ch. 11 - Account for the fact that tetrahydrofuran (THF) is...Ch. 11 - Prob. 11.14PCh. 11 - Write equations to show a combination of reactants...Ch. 11 - Propose a mechanism for this reaction.Ch. 11 - Prob. 11.17PCh. 11 - Prob. 11.18PCh. 11 - Prob. 11.19PCh. 11 - Prob. 11.20PCh. 11 - Ethylene oxide is the starting material for the...Ch. 11 - Prob. 11.22PCh. 11 - Predict the structural formula of the major...Ch. 11 - The following equation shows the reaction of...Ch. 11 - Propose a mechanism to account for this...Ch. 11 - Acid-catalyzed hydrolysis of the following epoxide...Ch. 11 - Prob. 11.27PCh. 11 - Prob. 11.28PCh. 11 - Prob. 11.29PCh. 11 - Propose a mechanism for the following...Ch. 11 - Show reagents and experimental conditions to...Ch. 11 - Starting with cis-3-hexene, show how to prepare...Ch. 11 - Show reagents to convert cycloheptene to each of...Ch. 11 - Show reagents to convert bromocyclopentane to each...Ch. 11 - Prob. 11.35PCh. 11 - Starting with acetylene and ethylene oxide as the...Ch. 11 - Following are the steps in the industrial...Ch. 11 - Prob. 11.38PCh. 11 - Prob. 11.39PCh. 11 - Aldehydes and ketones react with one molecule of...Ch. 11 - Prob. 11.42PCh. 11 - Write the products of the following sequences of...Ch. 11 - Using your reaction roadmap as a guide, show how...Ch. 11 - Using your reaction roadmap as a guide, show how...Ch. 11 - Using your reaction roadmap as a guide, show how...Ch. 11 - During the synthesis of the antiasthmatic drug...Ch. 11 - Prob. 11.48P
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- C) provide the drawing of stereochemical model explaining the obtained stereochemistry of product here C) provide the drawing of stereochemical model explaining the obtained stereochemistry of product herearrow_forward(a) What product is formed when each compound undergoes a thermal electrocyclic ring opening? (b) What product is formed when each compound undergoes a photochemical electrocyclic ring opening?arrow_forwardRank the compounds in each set from most to least reactive in an EAS reactionarrow_forward
- Draw the structure of the major product. If the answer for the product is a mixture of enantiomers draw one of the enantiomers and write "+ enantiomer." If the answer is a mixture of diaseteromers, draw all of them. Show arrow pushing mechanism and provide reasoning for each.arrow_forwardDraw the products of each reaction carried out under high-dilution conditions. Indicate the stereochemistry at all stereogenic centers.arrow_forwardDraw all products, including stereoisomers, in each reaction.arrow_forward
- Draw the major resonance structure for any resonance stabilized carbocations and label major onearrow_forwardDraw the major organic product of the reaction. Ignore stereochemistryarrow_forwardCyclooctatetraene is a not planar because it disrupts conjugation and avoids anti-aromaticity. b planar because it ensures conjugation and allows anti-aromaticity. c not planar because it disrupts conjugation and avoids aromaticity. d planar because it ensures conjugation and allows aromaticity.arrow_forward
- Rank the following carbocations in each set from most stable to least stable:arrow_forwardFor each of the following draw a constitutional isomer of the most favorable product(s).Note: Some products will have tetrahedral stereocenters. Indicate them with an asterisk(*), but, there is no need to draw all the strereoisomers for this exercise. Focus on the constitutional isomers.arrow_forwardPlease explain it. For each stereoisomer, draw the interconversion of chairs and identify the most stable. Give handwritten answer. ASAParrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning
