Q: Check Your Understanding on Crystal Structure 1. Differentiate crystal structure, crystal system, an...
A: 1. Crystal structure is made up of unit cells (repeating unit) and crystal structure is defined as ...
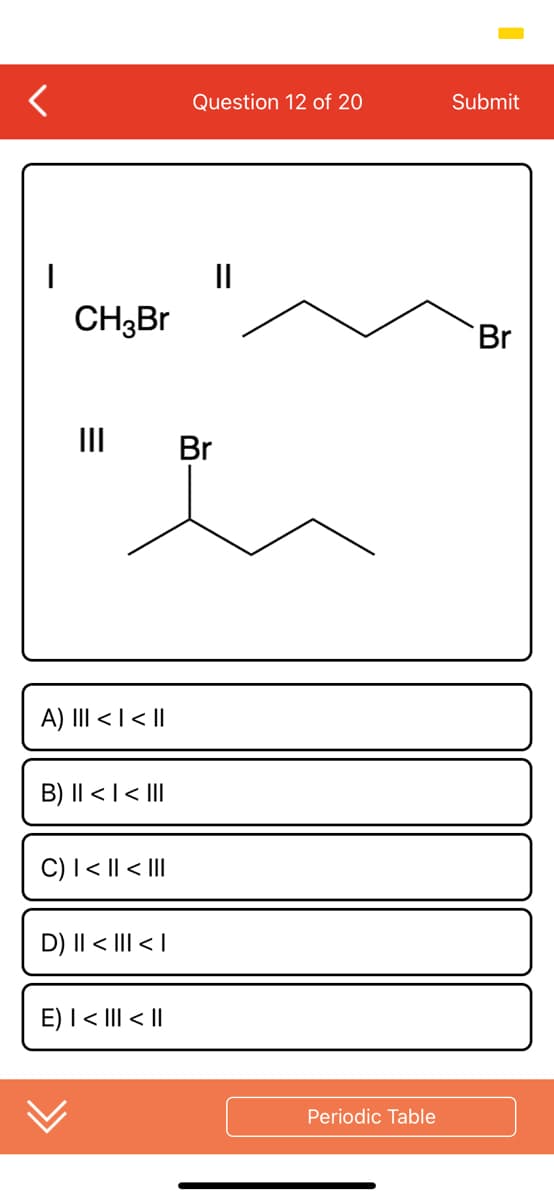
Q: 2/5 Liquid hexane (CH3(CH2)¸CH3) reacts with gaseous oxygen gas (0,) to produce gaseous carbon dioxi...
A:
Q: Which one of the following statements regarding Henry's law is not correct? * The partial pressure o...
A: Different gases have different Henry constant values at the same temperature. This means that it is ...
Q: A student prepares a stock solution of BaF2 by dissolving 50.00 g of a BaF2 in 1.00 L of water. An a...
A:
Q: Question 3 As the temperature of a reaction is increased, the rate of the reaction increases because...
A: As the temperature of a reaction increases, the rate of the reaction increases because the ......?.....
Q: The following reaction sequence is described as an introductory organic chemistry laboratory experim...
A: In synthetic organic chemistry, the combination of appropriate reactions under suitable conditions o...
Q: y grams
A:
Q: Calculate the pH of a solution with a hydrogen ion concentration of 4.5×10 M. a. Is this solution an...
A: pH = -log[H+] [H+] = 10-pH
Q: The symbol for rate constant is? Select one: a. L b. C c. k d. s
A: In chemical kinetics, the reaction rate constant or reaction rate coefficient is used as a proportio...
Q: How do we keep relative supersaturation low by controlling solute concentration?
A: ANSWER:-Option-3 is correct. 3.Concentration should be lower.
Q: What mass of Cu(NO 3) 2 can be prepared from the reaction of 5.00 grams of copper with 5.00 grams of...
A:
Q: Given the following information: 2 A(g) + B(g) = A2B(g) K1 = 5.0 x 10^4 2 A(g) + C2 (g) = 2 AC(g) K2...
A:
Q: nder certain conditions the rate of this reaction is zero order in ammonia with a rate constant of 0...
A: Given reaction is followed zero ordered reaction
Q: Is the precision of your two trials within 5 %?. Support your answer with calculations
A: In order to analyze the precision of an experiment, we calculate the Percent relative standard devia...
Q: Dr. Albert Apale, a well known Chemist, tested Random samples in the laboratory. The compounds were ...
A: Compounds are classified as : Ionic Saturated with 6 carbon molecules Unsaturated with 6 carbon mo...
Q: Given the following data for the NH4* +NO2 - N2 + 2H20 reaction Trial [NH4*] 0.015 M 0.030 0.015 Rat...
A:
Q: Solve all parts otherwise I will downvote
A: The required answers are given below The major product of the SN1 reaction is compound M. The major...
Q: The unit cell for tin has tetragonal symmetry, with a and blattice parameters of 0.5 and 0.318 nm, r...
A:
Q: Consider a buffer solution that contains 0.25 M CgHalCO2H){CO2K) and 0.15 M COH4(CO2K)2. pk (CH4(CO2...
A:
Q: Arrange the following in increasing melting points. Myristic acid, palmitic acid, oleic acid, steari...
A: Melting point of a substance is defined as a temperature at which solid changes into liquid.
Q: Which of the following statements about a gas chromatogram is correct? O Tailing indicates that the ...
A: A question based on analytical separations that is to be accomplished.
Q: 1. Provide a systematic name for each of the following compounds
A: Given; structure of molecules
Q: Under certain conditions the rate of this reaction is zero order in ammonia with a rate constant of ...
A: Given, 2NH3(g) ➝ N2(g) + 3 H2(g) Reaction is a zero order reaction. Rate constant (k) = 0.0091 M.s-1...
Q: A student prepares a stock solution of BaF2 by dissolving 50.00 g of a BaF2 in 1.00 L of water. An a...
A:
Q: + НОCH3 + H20 HO OCH3 H3CO OCH3 This reaction is classified as: addition substitution elimination cy...
A:
Q: What will be the major organic product from the dehydration of 2-butanol in the presence of a strong...
A: Correct option is (D) 2-butene explanation is given below
Q: Try to create a sample word problem about Hess’s Law. Show solution.
A: Hess law tells about the heat change takes place in a chemical reaction.
Q: Chromate-dichromate system 1. Prepare a clean spot plate and label four wells. Place 5 drops of 0. 1...
A: In a reversible reaction, the reaction takes place in forward direction as well as reverse direction...
Q: 41) A teenage child in Wisconsin throws a chunk of sodium metal into a lake consisting of freshwater...
A: Given: mass of sodium = atomic number of fluorine = 9 kg = 9000 g density of water = 1.01 g/mL heigh...
Q: pls solve this ques with a complete explanation within 10-15 minutes I'll give u multiple likes
A: To identify which type of reaction is given here.,
Q: 3) A solution has a pOH of 3.62. a. Is the solution an acid or a base?. Part b below first.) (If you...
A: As part of our community guidelines, we can answer only the first three sub-parts of the given quest...
Q: 1. Show all pertinent solutions neatly, orderly and completely. Indicate the oxidation state of the ...
A: Oxidation state can be defined as the total number of electrons that an atom either lose or gain fo...
Q: Which structural feature of Tryptophan and Tyrosine allows to absorb UV light? Is Aromatic structure...
A: Since precise protein quantitation is vital for all examinations connected with protein studies, var...
Q: 1.27 Draw the structures of the products that are formed when the following disulfide is reduced: CH...
A: Reduction can be defined as Addition of hydrogen to the substrate or removal oxygen from the substra...
Q: C4 H,0 8 4. 3. PM-00-190 ppm 00
A:
Q: Calculate A E for the reaction below at 25 *c. Sio 2(8) + 4HF(aq) SIF al0) + 2H 20 (1) -010.0 -320.8...
A:
Q: Mechanism 3. When heated the cyclobutene below coverts to a mixture of products A and B. The ftirst ...
A: Cyclobutene system is very unstable system as small ring containing double bond .Thus it faces huge ...
Q: H2N- CH-C- NH- CH2-C-NH-CH2- NH-CH-C-NH-CH-C-OH CH2 CH2 CH2 CH Compound A (Leu-enkephalin) H3C CH3 O...
A: Since you have asked multiple questions we will answer the first one for you. To get the remaining q...
Q: Identify the isomeric relationship between the molecules of each pair depicted below. Your choices ...
A:
Q: Density of Mole fraction of Molarity Molality % by Mass ppm solution solute
A:
Q: . What factors can affect a solute's Rf value in thin-layer chromatography? 2. Why is it necessary ...
A: Answer - According to the question - Rf values and reproducibility can be affected by a number of di...
Q: Discuss balancing chemical equation.
A: In a chemical reaction, the combination of appropriate reactants in a suitable molar ratio furnishes...
Q: the boiling point of a 6.95M solution of C12H22O11 in water? A. 100.0°C B.
A: Molarity = 6.95 M For very dilute solution, Molarity = Molality Using the Coligative property, □T...
Q: A student prepares a stock solution of BaF2 by dissolving 50.00g of a BaF2 in 1.00L of water. An add...
A: Given data,Mass of BaF2=50.00gVolume of water=1.00LMolarity of desired solution=0.100MVolume of desi...
Q: 2. Complete the following chemical equation below. Provide the reagent that will separate each pair ...
A: The group reagent of 3 rd group is ammonium sulphide solution or hydrogen sulphide gas in the presen...
Q: Calculate the standard enthalpy of formation of phenol, C 6H50H, at 298.15 K given that, at this tem...
A: Change of enthalpy of reaction is difference between total enthalpy of products and total enthalpy o...
Q: The correct IUPAC name for CH3-CH-CH=CH-CH3 CH3
A: Rule of IUPAC- 1) Longest chain as parent chain. 2) Numbering start from those side where more prior...
Q: Net Ionic Equation (for positive test result) Test Reagent Observations 2+ 0. 10 M K¸FE(CN), 3+ Fe 0...
A: Test Reagent Ion: Fe2+ Reagent: 0.10 M K3Fe(CN)6 Observation: A blue precipitate is formed Net Ion...
Q: EQUATION WRITING: Balance the following redox reactions. Use change in oxidation state method (oxida...
A: Let us step by step solve this:
Q: Consider the following rate law expression: rate = k[A][B]. Which of the following is not true about...
A:
Step by step
Solved in 2 steps with 1 images

- You finish doing an experiemnt with Benzoin. These are the results you get:Boiling start point: 137 ceciusBoiling end point: 130 ceciusVile while empty (with cap on): 15.348Vile with crude prod (with cap on: 15.748 Your starting weight: 1.00115.748 - 15.348 = 0.400 / 1.001 = 0.3996 or 39.96% YieldAnswer the following questions with this information:Initial Mass of impure sample:Mass of recrystallized benzoin:Percent reccovery of Benzoin:Melting pot range of purified benzoin:Literature melting point for benzoin:avg concentration: 8.937x10^-7Show that CV,vib goes to nR as T ∞.
- What mass of sucrose, C12H22O11, must be dissolved per liter of water (d= 0.998 g/ml) to obtain asolution with 2.75 mole percent C12H22O11?t Please don't provide hand writting solutionCalculate the volume of hydrogen at 0°C and 1.00 atmthat is required to convert 500.0 g linoleic acid (C18H32O2)to stearic acid (C18H36O2). (See Sec. 9.1.)
- A 5.000-g soil sample was analyzed for potassium content by extracting the potassium using 10.00 mL aqueous ammonium acetate solution. Following the extraction, the soil was filtered and rinsed. The filtrate with rinsings was diluted to exactly 50.00 mL. Then, 1.00 mL of this solution was diluted to 25.00 mL, and this dilution was tested with an instrument. The concentration in this 25.00 mL was found to be3.18 ppm. What is the concentration of the potassium in the soil in ppm?In the preparation of the standard curve for protein analysis, 50 mn BSA (bovine serum albumin) dissolved in H2O to a final volume of 5 mL was used as stock solution. What is the weight of BSA in 0.1 mL of stock solution? In 0.2 mL?Please don' tprovide hand writtin solution...



