11.11 Ethers can often be prepared by SN2 reaction of alkoxide ions, RO", with alkyl halides. Which of the two possible routes would be the better choice to synthesize the molecule shown below? Explain briefly. (a) (b) or + CH31 + CH3O™
11.11 Ethers can often be prepared by SN2 reaction of alkoxide ions, RO", with alkyl halides. Which of the two possible routes would be the better choice to synthesize the molecule shown below? Explain briefly. (a) (b) or + CH31 + CH3O™
Chapter14: Conjugated Compounds And Ultraviolet Spectroscopy
Section14.SE: Something Extra
Problem 50AP: -Ocimene is a pleasant-smelling hydrocarbon found in the leaves of certain herbs. It has the...
Related questions
Question
Ethers can often be prepared by SN2 reaction of alkoxide ions, RO*, with
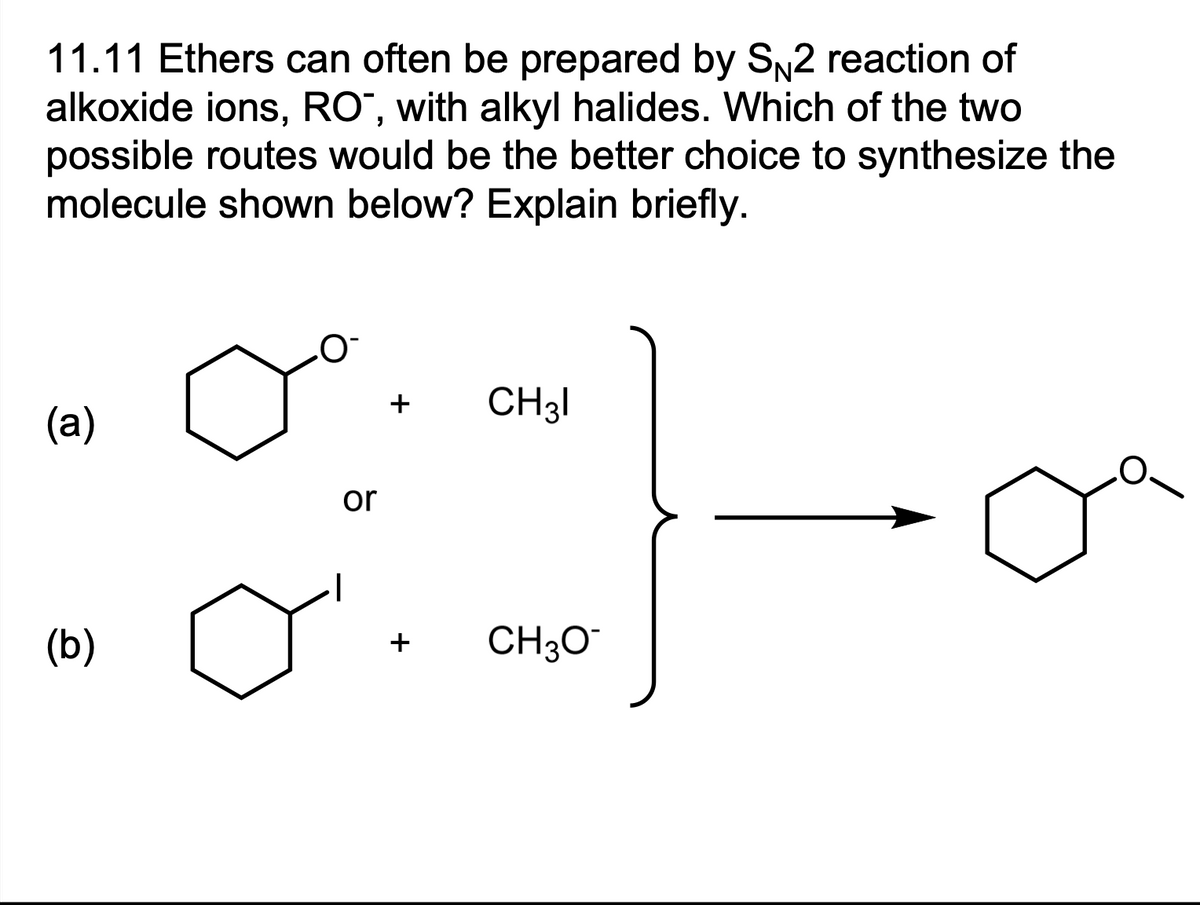
Transcribed Image Text:11.11 Ethers can often be prepared by SN2 reaction of
alkoxide ions, RO, with alkyl halides. Which of the two
possible routes would be the better choice to synthesize the
molecule shown below? Explain briefly.
(a)
(b)
O
or
+
CH31
+ CH3O
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 3 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

