
Answer the following questions about alcohol A.
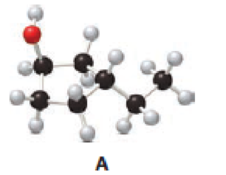
- a. Give the IUPAC name.
- b. Classify the alcohol as 1°, 2°, or 3°.
- c. Draw the products formed when A is dehydrated with H2SO4.
- d. What product is formed when A is oxidized with K2Cr2O7?
- e. Draw a constitutional isomer of A that contains an OH group.
- f. Draw a constitutional isomer of A that contains an ether.
a.
Interpretation:
For the below compound IUPAC name has to be determined.
Concept introduction:
Nomenclature of alcohol:
Firstly, find the longest carbon chain, which is bonded with the
Explanation of Solution
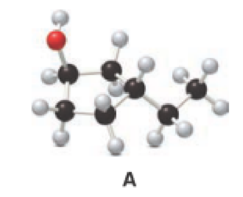
Given compound A is,
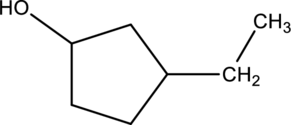
Step 1: In the given compound, black, white and red color balls indicates carbon, hydrogen and oxygen atoms respectively. Firstly, the ring which, is attached to the
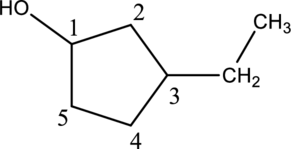
Step 2: For naming the compound, firstly, the parent carbon chain should be checked, and as it possess five membered ring, it is named as cyclopentane. Due to
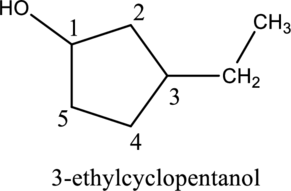
b.
Interpretation:
From the compound A, the type of alcohol has to be determined.
Concept introduction:
Classification of alcohol:
Generally, the alcohol group is bonded with minimum one alkyl group. The alcohol group is classified into three types, such as primary
Explanation of Solution
In the below compound A,
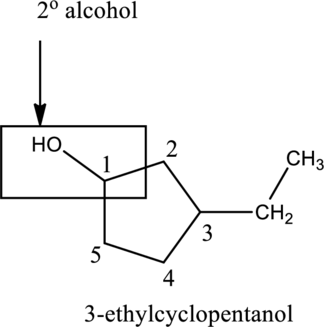
c.
Interpretation:
The product obtained when the compound A reacts with H2SO4 reagent has to be determined.
Concept introduction:
Dehydration of alcohol:
When alcohol reacts with
Explanation of Solution
Given stating material A is 3-ethylcyclopentanol
When 3-ethylcyclopentanol reacts with
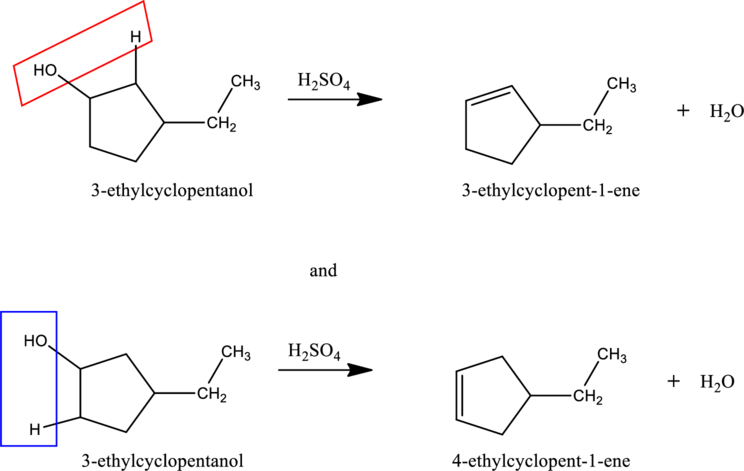
d.
Interpretation:
The product obtained when the compound A reacts with K2Cr2O7 reagent has to be determined.
Concept introduction:
Oxidation of alcohol:
During oxidation of alcohol, the number of
Explanation of Solution
Given stating material A is 3-ethylcyclopentanol
When 3-ethylcyclopentanol, the secondary alcohol containing one hydrogen atom on the carbon attached with OH group reacts with K2Cr2O7, it gets oxidized to 3-ethylcyclopentanone as shown below.
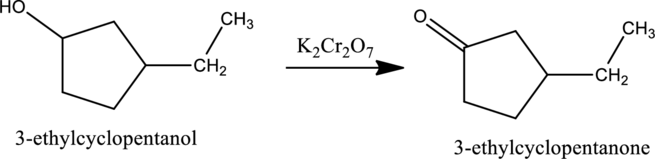
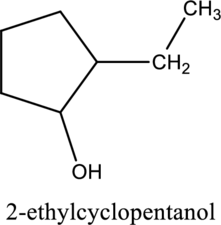
e.
Interpretation:
The constitutional isomer of compound A that contain
Concept introduction:
Constitutional isomers:
The compounds, that contain same molecular formula but different with respect to molecular orientation. In other words compounds having similar molecular formula and different structure are called as Constitutional isomers.
Explanation of Solution
In the given compound A molecular formula is
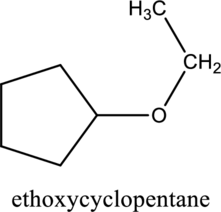
f.
Interpretation:
The constitutional isomer of compound A that contain an ether group has to be determined.
Concept introduction:
Refer to part ‘e.’.
Explanation of Solution
In the given compound A molecular formula is
Want to see more full solutions like this?
Chapter 12 Solutions
Principles of General, Organic, Biological Chemistry
- Identify the IUPAC name of the given structure. A. 2 - methylhexan-5-one B. 5 - methylhexan-2-one C. 2 - heptanone D. 5 - heptanone Identify the IUPAC name of the given structure. A. 4 - bromopentan-3-one B. 1 - bromobutan-2-one C. 2 - bromobutan-one D. None of the abovearrow_forwardGive the IUPAC name for each nitrile.arrow_forwardGlycerol contains: a. oxygens which are each bonded to two alkyl groups b. oxygens single-bonded to primary and secondary carbons c. Oxygens double-bonded to carbon, with alkyls on both sides d. Oxygens double-bonded to carbon, with alkyls on one side only e. Oxygens double-bonded to carbon, with an alkyl on one side and an --OH on the other sidearrow_forward
- Give an IUPAC or common name for each compound.arrow_forwardClassify each alcohol labeledarrow_forwardDraw the molecule that corresponds to each IUPAC name. (a) 2,3-dimethylcyclopentanone; (b) 4,4-difluoroheptanal;(c) 1,1,1-trichloropentan-3-one; (d) (1S,3S)-3-ethoxycyclohexanecarbaldehydearrow_forward
 Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781285199030Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781285199030Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning

