
Organic Chemistry
8th Edition
ISBN: 9781305580350
Author: William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 12, Problem 12.7P
Following are infrared spectra of 2-methyl-1-butanol and tert-butyl methyl ether. Assign each compound its correct spectrum.
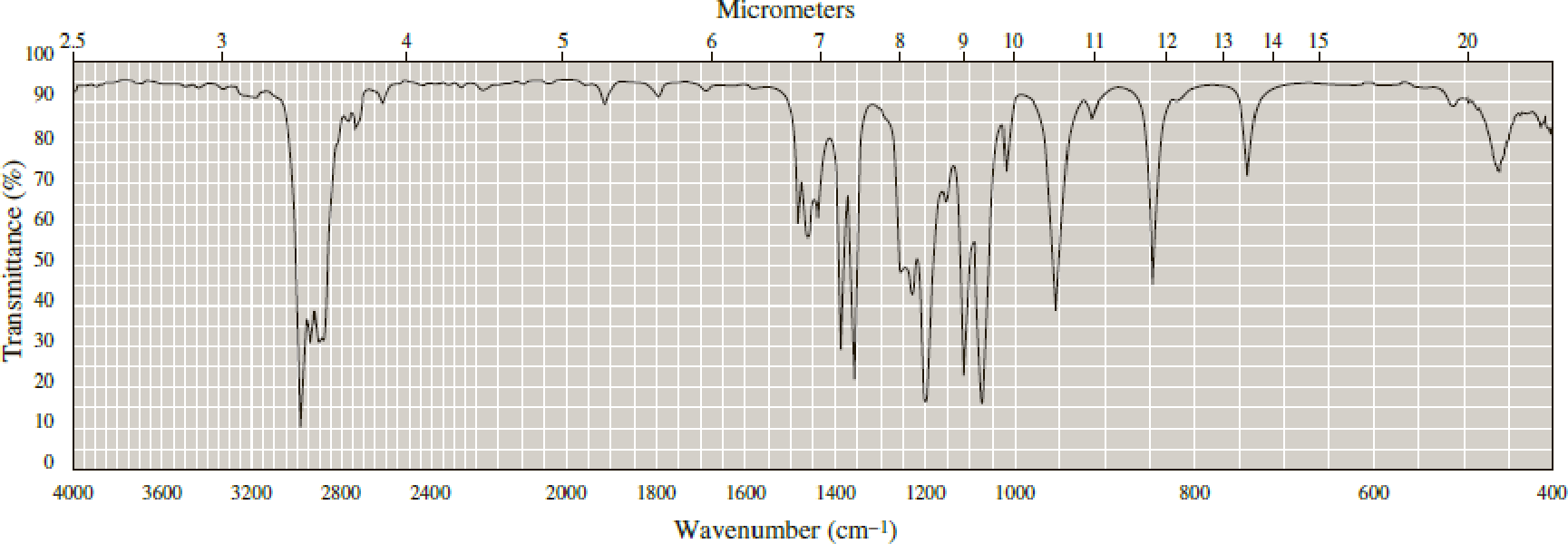
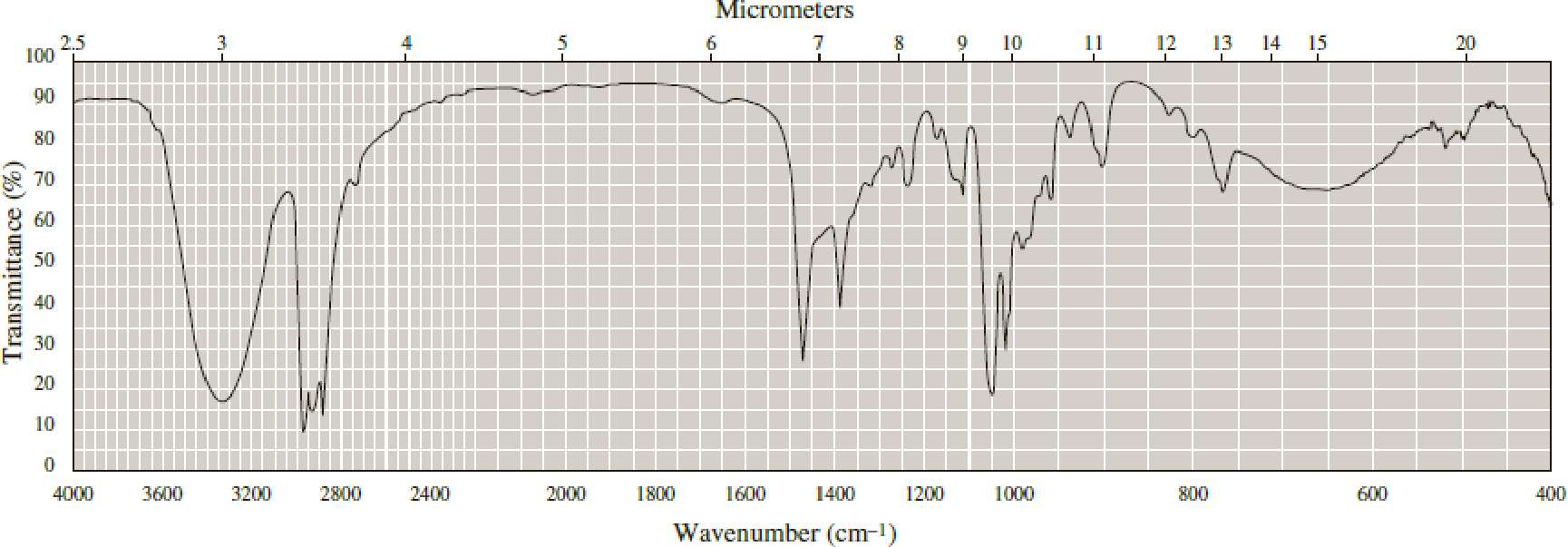
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
Predict the characteristic infrared absorptions of the functional groups in the following molecules. ) pentan-2-ol
Predict the characteristic infrared absorptions of the functional groups in the following molecules. ) diethylamine
Predict the characteristic infrared absorptions of the functional groups in the following molecules. pentanoic acid
Chapter 12 Solutions
Organic Chemistry
Ch. 12.3 - Prob. 12.1PCh. 12.3 - Prob. 12.2PCh. 12.3 - A compound shows strong, very broad IR absorption...Ch. 12.3 - Propanoic acid and methyl ethanoate are...Ch. 12 - Following are infrared spectra of...Ch. 12 - Following are infrared spectra of nonane and...Ch. 12 - Following are infrared spectra of...Ch. 12 - The IR CC stretching absorption in symmetrical...Ch. 12 - Prob. 12.9PCh. 12 - A compound has strong infrared absorptions at the...
Additional Science Textbook Solutions
Find more solutions based on key concepts
Determine the number of protons, neutrons, and electrons in the following atoms: a. a hydrogen atom that has a ...
General, Organic, and Biological Chemistry (3rd Edition)
Give one example from main group chemistry that illustrates each of the following descriptions: (a) Covalent ne...
General Chemistry: Atoms First
What is the pH range for acidic solutions? For basic solutions?
EBK INTRODUCTION TO CHEMISTRY
The smallest building blocks inside your cell phone are about 1000 times smaller than the diameter of a human h...
Chemistry In Context
The structural formula of 1, 2-dimethylbenzene needs to be drawn. Concept introduction: The ring structures of ...
Chemistry: Matter and Change
Calculate the lattice energy of CaCl2 using a Born-Haber cycle and data from Appendices F and L and Table 7.5. ...
Chemistry & Chemical Reactivity
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Following are infrared spectra of nonane and 1-hexanol. Assign each compound its correct spectrum.arrow_forwardFollowing are mass spectra for the constitutional isomers 2-pentanol and 2-methyl- 2-butanol. Assign each isomer its correct spectrum.arrow_forward2. A. Match the following IR spectra with their respective molecules butane, 1-butene, and 1- butyne. Explain your answer. B. Assigned the major peaks in spectra (indicate the positions of vibrational modes belonging to the major functional groups in molecules). C. Calculate the number of vibrational modes in butane.arrow_forward
- In infrared spectra, the O-H vibrational frequency of alcohols occurs as a broad peak (peak absorbable) in the region 3200-3600 cm -1, true or false?arrow_forwardHow could you distinguish between methyl butanoate and pentanoic acid by infrared spectroscopy?arrow_forwardOrganic Chemistry - How many signals would you expect in the 1H NMR spectrum of HOCH2CH2CH2CH2OH?arrow_forward
- I need help interpreting this IR spectrum of cyclohexanone. I'm not looking for anything in general just a description and explanation of what can be observedarrow_forwardHow many unique 13C NMR and 1H NMR signals exist in the spectrum for the compound: 1,3-dibromobenzene?arrow_forwardPredict the characteristic infrared absorptions of the functional groups in the following molecules.(a) cyclohexenearrow_forward
- Your classmate needs to decide if the spectrum shown below is a 13CNMR or an 1HNMR spectrum of 2-methyl propane. What is your advice to the student and why?arrow_forwardThe 13C NMR spectrum of a compound with molecular formula C7H16O, measured in CDCl3. Assign the carbons by putting the appropriate letter over each peak in the spectrumarrow_forwardPredict the number of peaks and their multiplicity in the off-resonance decoupled CMR spectra of the following compounds : (a) p-Dichlorobenzene (c) 1,4-Dioxane (b) p-Xylene (d) Methyl cyclopropanearrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:9781305079113
Author:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:Cengage Learning
IR Spectroscopy; Author: Professor Dave Explains;https://www.youtube.com/watch?v=_TmevMf-Zgs;License: Standard YouTube License, CC-BY