
Concept explainers
Write structural formulas and give the IUPAC names for all the isomers of
Interpretation:
The structural formulas and IUPAC names for all the isomers of
Concept introduction:
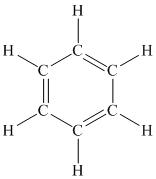
A benzene ring is a cyclic six membered ring containing single and double bonds alternately between carbon atoms. One hydrogen atom is attached to each carbon atom.
In a mono substituted benzene ring, one hydrogen atom is replaced by a substituent.
The substituent should be such that each carbon atom must have formed four bonds and other atoms, if any, should have complete octets.
Answer to Problem 33P
Solution:
Explanation of Solution
The structure of a benzene ring is as follows:
One hydrogen atom is replaced by the group
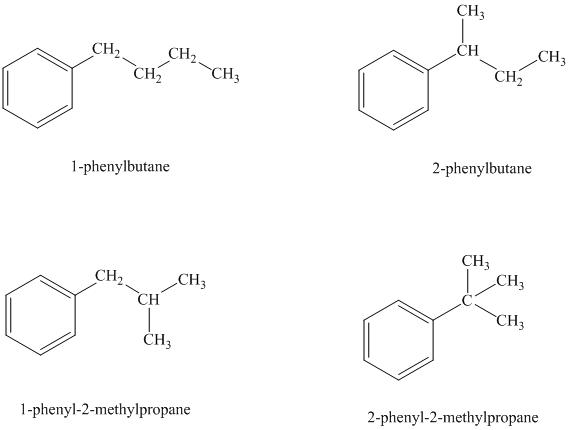
There are four possible ways in which this group can be attached to the ring.
The structures and IUPAC names of the isomers are shown below:
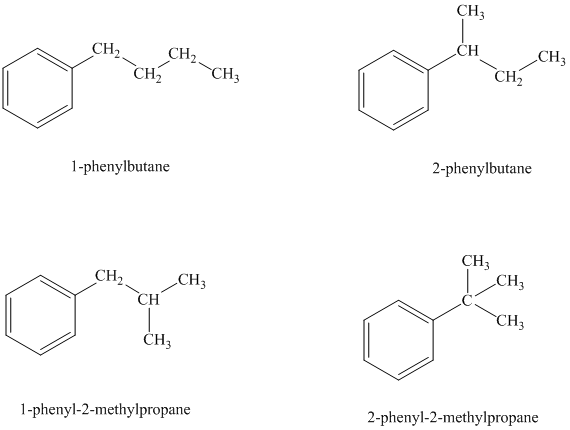
In a mono substituted benzene ring, one hydrogen atom is substituted by a substituent. An alkyl substituent can be arranged such that each carbon atom must have formed four bonds and other atoms if any should have complete octets.
Want to see more full solutions like this?
Chapter 12 Solutions
Connect Access Card Two Year for Organic Chemistry
- trans-Cyclodecene boils at 193C, but cis-cyclodecene boils at 195.6C. Write structural formulas for these two compounds.arrow_forwardWrite the structure of the molecule of a compound whose IUPAC name is 1-phenylpropan-2-ol.arrow_forwardWrite the structural formula for 3-methylcyclohexanol.arrow_forward
- Give A Summary of Substitution and Elimination Reactions of Alcohols ?arrow_forwardWhat is the structure of a compound with the formula C7H14O that has an ether and cyclobutane ring, including its IUPAC name? In addition, what is the structure of the compound with the formula C7H14O that does NOT contain the functional groups: epoxide, ether, cycloalkane, alcohol, alkene. Indicate the functional groups used that are present in the structure made for this compound.arrow_forwardWrite down the 5 different types of formulae used in organic chemistry for the compound ethene.arrow_forward
- What are some of the ways that the features of carbon-tocarbon bonds influence the stability and 3-D structure of organic molecules?arrow_forwardWrite a balanced reaction equation for the synthesis of 2- butoxynaphthalene. Include correctly drawn structures for any and all organic reactants and products.arrow_forwardcondense structural formulA of C4H8O with carbon-carbon double bonded and a hydroxylic grouparrow_forward
- What is the structural difference between primary, secondary, and tertiary alcohols?arrow_forwardWrite structural formulas for each of the possible isomers ofn-pentyne that are formed by moving the position of the triplebond.arrow_forwardgive iupac name and Classify the following compounds as to what organic group it belongs. Choices: alkane, alkene, alkyne, arene, alcohol, phenol, ether, aldehyde, ketone, thiol.If it is an alcohol indicate the type/classification.arrow_forward
 World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning




