
Concept explainers
(a)
Interpretation:
An efficient synthesis that can be used to achieve each of the following transformations is to be determined.
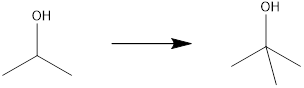
Concept introduction:
The product molecule has one more carbon atom than the starting material. For the generation of tertiary alcohol from secondary alcohol, there must be oxidation of alcohol to the carbonyl group, after which, there must be an addition of a methyl fragment, preferably using a Grignard reagent. This after hydrolysis can yield the desired tertiary alcohol.
(b)
Interpretation:
An efficient synthesis that can be used to achieve each of the following transformations is to be determined.

Concept introduction: The final product has one more carbon atom than the starting material. This can be facilitated by the addition of a methyl group, preferably using a Grignard reagent after conversion of
(c)
Interpretation:
The efficient synthesis that can be used to achieve the following transformation is to be determined.
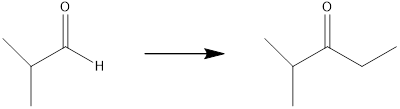
Concept introduction:
The final product contains two additional carbon atoms, so the synthesis requires a carbon-carbon bond-forming reaction. To facilitate this, the desired product should be produced by oxidizing the generated alcohol using the proper Grignard reagent.
(d)
Interpretation:
The efficient synthesis that can be used to achieve each of the following transformations is to be determined.
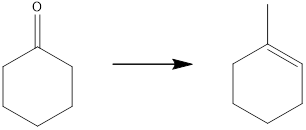
Concept introduction:
The target molecule contains one additional carbon atom, so the synthesis must have a carbon-carbon bond-forming reaction. This can be achieved by using a Grignard reagent followed by an elimination reaction which can yield the desired unsaturated product.
(e)
Interpretation:
The efficient synthesis that can be used to achieve each of the following transformations is to be determined.

Concept introduction:
The target molecule contains three additional carbon atoms, so the reaction needs a Carbon−Carbon bond-forming reaction. The
(f)
Interpretation:
An efficient synthesis that can be used to achieve each of the following transformations is to be determined.
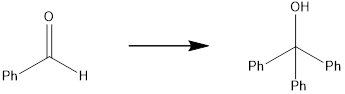
Concept introduction:
The target molecule has two additional phenyl groups, which can be done by adding Grignard reagent containing phenyl groups two times, followed by oxidation. The starting material contains carbonyl carbon which is the reaction center for the phenyl part of the Grignard reagent.
Want to see the full answer?
Check out a sample textbook solution
Chapter 12 Solutions
ORGANIC CHEMISTRY 2-SEMESTER-ACCESS
- Nonconjugated , -unsaturated ketones, such as 3-cyclohexenone, are in an acid-catalyzed equilibrium with their conjugated , -unsaturated isomers. Propose a mechanism for this isomerization.arrow_forwardWith the starting material provided, propose a synthesis pathway to prepare the diketone shown below. سلامarrow_forwardDevise a concise synthesis for the following transformations. Clearly show the reagent and productfor each step. Each synthesis can be accomplished in two stepsarrow_forward
- Please answer with full explanation.arrow_forwardUsing cyclohexanone as the starting material, describe how the following compounds can be synthesized:arrow_forwardProvide a detailed arrow-pushing mechanism for the following transformation: Me PHOH PhO. Et Мe Me Et Me OTs Provide a detailed arrow-pushing mechanism for the following transformation:arrow_forward
- When cis-2-decalone is dissolved in ether containing a trace of HCI, an equilibrium is established with trans-2-decalone. The latter ketone predominates in the equilibrium mixture. H H HCI cis-2-Decalone trans-2-Decalone Propose a mechanism for this isomerization and account for the fact that the trans iso- mer predominates at equilibrium.arrow_forwardCyclohexene can be converted to 1-cyclopentenecarbaldehyde by the following series of reactions. Propose a structural formula for each intermediate compound.arrow_forwardPropose an efficient synthesis for each of the following transformations:arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

