
College Physics
11th Edition
ISBN: 9781305952300
Author: Raymond A. Serway, Chris Vuille
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Textbook Question
thumb_up100%
Chapter 12.3, Problem 12.2QQ
Identify the paths A, B, C, and D in Figure 12.11 as isobaric, isothermal, isovolumetric, or adiabatic. For path B, Q = 0.
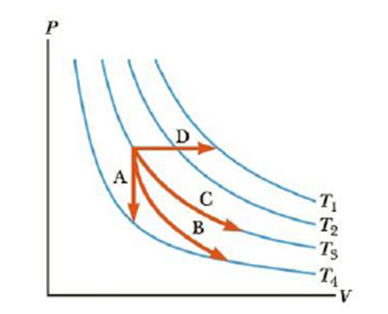
Figure 12.11 (Quick Quiz 12.2) Identify the nature of paths A, B, C, and D
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
please do fast
5. Find the expression for the entropy of a single harmonic oscillator.
ILLUSTRATE THE PROBLEM ONLY!!!
5 moles of cinnamaldehyde gas that has an initial volume of ten liters expands under the following conditions: 185°F and 800mmHg external pressure.
a. How much work is done? Express your answer in L atm.
b. Assuming that the container is cylindrical with a base radius of 10cm and can only expand vertically, how much kinetic energy does a single molecule possesses if it travels from base to base of the expanded cylinder in 10μs? Express your answer in J.
ILLUSTRATE THE PROBLEM ONLY!!!
Is the statement true of false? Support the answer
a. For every cyclic process, the final state of the system is the same as the initial state.
Chapter 12 Solutions
College Physics
Ch. 12.1 - By visual inspection, order the PV diagrams shown...Ch. 12.3 - Identify the paths A, B, C, and D in Figure 12.11...Ch. 12.4 - Three engines operate between reservoirs separated...Ch. 12.5 - Which of the following is true for the entropy...Ch. 12.5 - Prob. 12.5QQCh. 12 - Two identical containers each hold 1 mole of an...Ch. 12 - Which one of the following statements is true? (a)...Ch. 12 - Prob. 3CQCh. 12 - Prob. 4CQCh. 12 - For an ideal gas in an isothermal process, there...
Ch. 12 - An ideal gas undergoes an adiabatic process so...Ch. 12 - Is it possible to construct a heat engine that...Ch. 12 - A heat engine does work Weng while absorbing...Ch. 12 - When a sealed Thermos bottle full of hot coffee is...Ch. 12 - The first law of thermodynamics is U = Q + W. For...Ch. 12 - The first law of thermodynamics says we cant get...Ch. 12 - Objects A and B with TA TB are placed in thermal...Ch. 12 - Prob. 13CQCh. 12 - Prob. 14CQCh. 12 - An ideal gas is compressed to half its initial...Ch. 12 - A thermodynamic process occurs in which the...Ch. 12 - Prob. 17CQCh. 12 - An ideal gas is enclosed in a cylinder with a...Ch. 12 - Sketch a PV diagram and find the work done by the...Ch. 12 - Gas in a container is at a pressure of 1.5 atm and...Ch. 12 - Find the numeric value of the work done on the gas...Ch. 12 - A gas expands from I to F along the three paths...Ch. 12 - A gas follows the PV diagram in Figure P12.6. Find...Ch. 12 - A sample of helium behaves as an ideal gas as it...Ch. 12 - (a) Find the work done by an ideal gas as it...Ch. 12 - One mole of an ideal gas initially at a...Ch. 12 - (a) Determine the work done on a fluid that...Ch. 12 - A balloon holding 5.00 moles of helium gas absorbs...Ch. 12 - A chemical reaction transfers 1250 J of thermal...Ch. 12 - Prob. 13PCh. 12 - A cylinder of volume 0.300 m3 contains 10.0 mol of...Ch. 12 - A gas expands from I to F in Figure P12.5. The...Ch. 12 - In a running event, a sprinter does 4.8 105 J of...Ch. 12 - A gas is compressed at a constant pressure of...Ch. 12 - A quantity of a monatomic ideal gas undergoes a...Ch. 12 - A gas is enclosed in a container fitted with a...Ch. 12 - A monatomic ideal gas under-goes the thermodynamic...Ch. 12 - An ideal gas is compressed from a volume of Vi =...Ch. 12 - A system consisting of 0.025 6 moles of a diatomic...Ch. 12 - An ideal monatomic gas expands isothermally from...Ch. 12 - An ideal gas expands at constant pressure. (a)...Ch. 12 - An ideal monatomic gas contracts in an isobaric...Ch. 12 - An ideal diatomic gas expands adiabatically from...Ch. 12 - An ideal monatomic gas is contained in a vessel of...Ch. 12 - Consider the cyclic process described by Figure...Ch. 12 - A 5.0-kg block of aluminum is heated from 20C to...Ch. 12 - One mole of gas initially at a pressure of 2.00...Ch. 12 - A gas increases in pressure from 2.00 atm to 6.00...Ch. 12 - An ideal gas expands at a constant pressure of...Ch. 12 - A heat engine operates between a reservoir at 25C...Ch. 12 - A heat engine is being designed to have a Carnot...Ch. 12 - The work done by an engine equals one-fourth the...Ch. 12 - In each cycle of its operation, a heat engine...Ch. 12 - One of the most efficient engines ever built is a...Ch. 12 - A lawnmower engine ejects 1.00 104 J each second...Ch. 12 - An engine absorbs 1.70 kJ from a hot reservoir at...Ch. 12 - A heat pump has a coefficient of performance of...Ch. 12 - A freezer has a coefficient of performance of...Ch. 12 - Prob. 42PCh. 12 - In one cycle a heat engine absorbs 500 J from a...Ch. 12 - A power plant has been proposed that would make...Ch. 12 - Prob. 45PCh. 12 - A heat engine operates in a Carnot cycle between...Ch. 12 - A Styrofoam cup holding 125 g of hot water at 1.00...Ch. 12 - A 65-g ice cube is initially at 0.0C. (a) Find the...Ch. 12 - A freezer is used to freeze 1.0 L of water...Ch. 12 - What is the change in entropy of 1.00 kg of liquid...Ch. 12 - A 70.0-kg log falls from a height of 25.0 m into a...Ch. 12 - A sealed container holding 0.500 kg of liquid...Ch. 12 - Prob. 53PCh. 12 - When an aluminum bar is temporarily connected...Ch. 12 - Prepare a table like Table 12.3 for the following...Ch. 12 - Prob. 56PCh. 12 - Prob. 57PCh. 12 - Prob. 58PCh. 12 - Sweating is one of the main mechanisms with which...Ch. 12 - Prob. 60PCh. 12 - Suppose a highly trained athlete consumes oxygen...Ch. 12 - A Carnot engine operates between the temperatures...Ch. 12 - Prob. 63APCh. 12 - A Carnot engine operates between 100C and 20C. How...Ch. 12 - A substance undergoes the cyclic process shown in...Ch. 12 - When a gas follows path 123 on the PV diagram in...Ch. 12 - Prob. 67APCh. 12 - An ideal gas initially at pressure P0, volume V0,...Ch. 12 - One mole of neon gas is heated from 300. K to 420....Ch. 12 - Every second at Niagara Falls, approximately 5.00 ...Ch. 12 - A cylinder containing 10.0 moles of a monatomic...Ch. 12 - Prob. 72APCh. 12 - Suppose you spend 30.0 minutes on a stair-climbing...Ch. 12 - Hydrothermal vents deep on the ocean floor spout...Ch. 12 - An electrical power plant has an overall...Ch. 12 - A diatomic ideal gas expands from a volume of VA =...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, physics and related others by exploring similar questions and additional content below.Similar questions
- Consider a discrete random variable X with 2n+1 symbols xi, i = 1, 2, …, 2n+1. Determine the upper and lower bounds on the entropy when (a) p(x1)=0 (b) p(x1)=1/2arrow_forwardWhat are the key principles and applications of the adiabatic approximation in the field of physics?arrow_forward3. 0.5 moles of cinnamaldehyde gas that has an initial volume of ten liters expands under the following conditions: 185°F and 800mmHg external pressure.a. How much work is done? Express your answer in L atm.b. Assuming that the container is cylindrical with a base radius of 10cm and can only expand vertically, how much kinetic energy does a single molecule possesses if it travels from base to base of the expanded cylinder in 10μs? Express your answer in J. (Note that 1L atm=101.325J)arrow_forward
- Homework 6 Problem 6: A rigid tank of volume V = 0.015 m3 contains carbon monoxide at a temperature of T0 = 21° C and a pressure of P0 = 9.00 × 105 Pa. This molecule should be treated as a diatomic ideal gas with active vibrational modes. Part (a) Calculate the pressure of the gas in pascal at this increased temperature. P = ______ Part (b) Calculate the change to the internal energy of the gas in joules. ΔU = ______ Part (c) Calculate the change in the entropy of the gas in joules per kelvin. ΔS = ______arrow_forwardHow would you describe the internal energy of a system in terms of the kinetic and potential energy of all its particles and atoms?arrow_forwardAlso answer d ) For state 1 evaluate the specific volume assuming the steam behaves as an ideal gas and comment on your result.arrow_forward
- Determine the internal energy of compressed liquid water at 80°C and 5 MPa, using (a) data from the compressed liquid table and (b) saturated liquid data. What is the error involved in the second case?arrow_forwardCalculate the work required to move the 1μC test load, which is 50 cm from the center of the 20 cm conductive sphere with a load of 5 μC, to a point 10 cm from the center of the sphere. (k = 9 × 109 Nm2 / C2)arrow_forwarda) Calculate the work for an ideal gas that expands isothermally at T0 from V1 to V2.b) Carry out the Legendre transform of U on the two variables S and V.arrow_forward
- A rigid container contains a mixture of 2mols of a polyatomic gas at a temperature of 3000K. You then heat the mixture providing 39907.02J of heat. At the end of the process, the mixture is at a temperature of 3600K.If at 3000K the translational as well as the 3 rotational degrees of freedom of the polyatomic gas are already activated, how many vibrational degrees of freedom does it have activated in this temperature range? (Assume the number of degrees of freedom does not change during the process).arrow_forward3. 0.5 moles of cinnamaldehyde gas that has an initial volume of ten liters expands under the followingconditions: 185°F and 800mmHg external pressure.a. How much work is done? Express your answer in L atm.b. Assuming that the container is cylindrical with a base radius of 10cm and can only expandvertically, how much kinetic energy does a single molecule possesses if it travels from baseto base of the expanded cylinder in 10μs? Express your answer in J.arrow_forwardA small block of mass m = 230 g is released from rest at point , along the horizontal diameter on the inside of a hemispherical bowl of radius R = 29.9 cm, and the surface of the bowl is rough (see figure below). The block's speed at is 1.70 m/s. (a) What is its kinetic energy at point ? (b) How much mechanical energy is transformed into internal energy as the block moves from point to point ? (c) Is it possible to determine the coefficient of friction from these results in any simple manner? Explain your answer.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 College PhysicsPhysicsISBN:9781305952300Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning
College PhysicsPhysicsISBN:9781305952300Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning

College Physics
Physics
ISBN:9781305952300
Author:Raymond A. Serway, Chris Vuille
Publisher:Cengage Learning
The Second Law of Thermodynamics: Heat Flow, Entropy, and Microstates; Author: Professor Dave Explains;https://www.youtube.com/watch?v=MrwW4w2nAMc;License: Standard YouTube License, CC-BY