
(a)
Interpretation:
The alcohol should be identified when the given 2-methylpropanal undergoes reduction with sodium borohydrate.
Concept introduction:
NaBH4 (Sodium borohydride):
Sodium borohydride is used as a reducing agent.
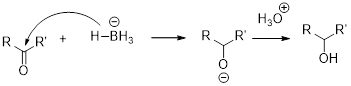
(b)
Interpretation:
The alcohol should be identified when the given cyclohexanone undergoes reduction with sodium borohydrate.
Concept introduction:
NaBH4 (Sodium borohydride):
Sodium borohydride is used as a reducing agent.
Aldehydes and ketons react with sodium borohydrate undergoes reduction to forms alcohols.
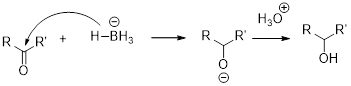
(c)
Interpretation:
The alcohol should be identified when the given tert-butylcyclohexanone undergoes reduction with sodium borohydrate.
Concept introduction:
NaBH4 (Sodium borohydride):
Sodium borohydride is used as a reducing agent.
Aldehydes and ketons react with sodium borohydrate undergoes reduction to forms alcohols.
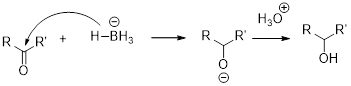
(d)
Interpretation:
The alcohol should be identified when the given methyl phenyl
Concept introduction:
NaBH4 (Sodium borohydride):
Sodium borohydride is used as a reducing agent.
Aldehydes and ketons react with sodium borohydrate undergoes reduction to forms alcohols.
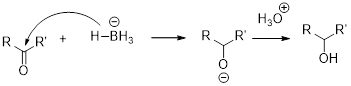
Want to see the full answer?
Check out a sample textbook solution
Chapter 12 Solutions
Essential Organic Chemistry (3rd Edition)
- Convert benzene into attached compound. You may also use any inorganic reagents and organic alcohols having four or fewer carbons. One step of the synthesis must use a Grignard reagent.arrow_forwardWhat happens when water, alcohols or other protic solvents are used in Grignard reactions?arrow_forwardWittig reactions with the following -chloroethers can be used for the synthesis of aldehydes and ketones. (a) Draw the structure of the triphenylphosphonium salt and Wittig reagent formed from each chloroether. (b) Draw the structural formula of the product formed by treating each Wittig reagent with cyclopentanone. Note that the functional group is an enol ether or, alternatively, a vinyl ether. (c) Draw the structural formula of the product formed on acid-catalyzed hydrolysis of each enol ether from part (b).arrow_forward
- Convert benzene into attached compound. You may also use any inorganicreagents and organic alcohols having four or fewer carbons. One step of the synthesis must use a Grignard reagent.arrow_forwardSynthetize 3-pentynal from 3-butynal using whatever organic/inorganic reagents are needed.arrow_forwardWhat would be the organic product for each reaction?arrow_forward
- Convert benzene into each compound. You may also use any inorganic reagents and organic alcohols having four or fewer carbons. One step of the synthesis must use a Grignard reagent.arrow_forwardWhy is it not advisable to use aqueous hydrochloric acid in a Grignard reaction of a ketone? A) The Grignard reagent will react with the acid and cannot react with the ketone. B) The ketone will be protonated and will become unreactive. C) The ketone will form an unreactive enol. D) The Grignard reagent won't dissolve in aqueous solutionsarrow_forwardConvert benzene into each compound. You may also use any inorganic reagents and organic alcohols having three carbons or fewer. One step of the synthesis must use a Grignard reagent.arrow_forward
- What happens when an aldehyde gives a silver mirror when treated with Tollen’s reagent and give red-brown precipitate when treated with Fehling’s solution? Explain how they are formed using chemical equations.arrow_forwardexplain why carbonyl compounds are so attractive to both nucleophiles and electrophilesarrow_forwardExplain Addition of Alcohols—Acetal Formation ?arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

