
In your own words, explain why an object that has more symmetry elements is said to have “higher symmetry” than an object with fewer symmetry elements.
Interpretation:
The reason as to why an object that has more symmetry elements is said to have “higher symmetry” than an object with fewer symmetry elements is to be stated.
Concept introduction:
A symmetry operation is defined as an action on an object to reproduce an arrangement that is identical to its original spatial arrangement. The spatial arrangement of the object remains identical after a symmetry operation. The point of reference through which a symmetry operation takes place is termed as a symmetry element.
Answer to Problem 13.1E
An object that has more symmetry elements is said to have “higher symmetry” than an object with fewer symmetry elements because the object with more symmetry element has more identical spatial arrangements.
Explanation of Solution
The plane of symmetry is a symmetry element that is present in an object that can be divided into two equal identical halves. For example, the letter H has two planes of symmetry. These planes of symmetry are represented as,
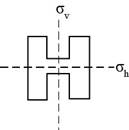
Figure 1
If the letter H is written upside down then it will look totally identical to its original arrangement.
The letter A has only one plane of symmetry. The plane of symmetry in letter A is represented as,
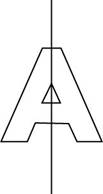
Figure 2
If the letter A is written upside down then it will not look identical to its original arrangement.
The symmetry element of letter H is more than letter A and letter H is more symmetrical than letter A.
Therefore, it can be concluded that the object with more symmetry elements has more identical spatial arrangements. An object that has more symmetry elements is said to have “higher symmetry” than an object with fewer symmetry elements.
The object with more symmetry element has more identical spatial arrangements. Therefore, an object that has more symmetry elements is said to have “higher symmetry” than an object with fewer symmetry elements.
Want to see more full solutions like this?
Chapter 13 Solutions
Physical Chemistry
- Identify all the symmetry elements present in the tetrahedron, the cube, and the octahedron.arrow_forwardIdentify the symmetry elements present in the following objects. a The Eiffel Tower. You may have to look up a picture of it if you dont remember its shape b Any book ignore the printing. c An octagonal wood block. d A jack from the set of jacks pictured here: Note that some of the points end differently.arrow_forwardIdentify the symmetry elements present in the following objects. a A ream of blank paper, no holes. b A ream of blank three-holed paper. c A round pencil, unsharpened, with cylindrical eraser. d A round pencil, sharpened, with cylindrical eraser.arrow_forward
- Determine which single symmetry operation of the following point groups is equivalent to the given combination of multiple symmetry operations. a In C2v, C2v=? b In C2h, iC2=? c In D6h, C6h=? d In D2d, C2C2=? e In Oh, iS4=?arrow_forwardThe Cu(H2O)62+ complex has octahedral symmetry. Is a transition from a T2g state to an Eg state allowed if the transition moment operator has a T1u symmetry label?arrow_forwarda In the Td point group, an S41 improper rotation is equivalent to what other improper rotation? b In the D6h point group, the symmetry operation labeled C21 is equivalent to what other symmetry operation?arrow_forward
- Linearly polarized light can be assigned a specific irreducible representation of a symmetry point group. If the electronic ground state of methane has A1g symmetry and x- polarized light has the label T2, what are the symmetry labels of allowed excited electronic states? Use the Td character table in Appendix 3.arrow_forwardAssume that x- polarized light can be assigned an E symmetry species in a system that has C4v symmetry. Can a transition from E state to a B2 state occur? Why or why not?arrow_forwardLinearly polarized light can be assigned a specific irreducible representation of a symmetry point group. If the electronic ground state of benzene has A1g symmetry and y- polarized light has the label E1u, what are the symmetry labels of allowed excited electronic states? Use the D6h character table in Appendix 3.arrow_forward
- a Unlike methane, bromochlorofluoromethane (CHBrClF) is chiral. Determine all symmetry elements that are present in CHBrClF and identify its point group. b If the fluorine in this molecule were substituted with a hydrogen atom, what is the point group for the new molecule? Is it chiral?arrow_forwardWithout using the great orthogonality theorem, reduce the given irreducible representation in C2v symmetry. Does your answer make sense? EC25555arrow_forwardConstruct the symmetry-adapted linear combination molecular orbitals for hydrogen sulfide, H2S.arrow_forward
 Physical ChemistryChemistryISBN:9781133958437Author:Ball, David W. (david Warren), BAER, TomasPublisher:Wadsworth Cengage Learning,
Physical ChemistryChemistryISBN:9781133958437Author:Ball, David W. (david Warren), BAER, TomasPublisher:Wadsworth Cengage Learning, Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

