
The reaction X → Y shown here follows first-order kinetics. Initially different amounts of X molecules are placed in three equal-volume containers at the same temperature. (a) What are the relative
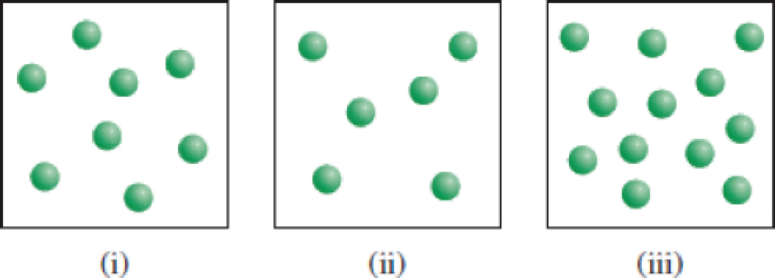
(a)
Interpretation:
The relative rate of the reaction in the given three containers has to be determined.
Concept introduction:
Rate of the reaction is the change in the concentration of reactant or a product with time.
Rate equation for the general reaction
Explanation of Solution
The given first order reaction is,
For first order reaction,
X molecules are placed in three equal-volume containers at the same temperature.
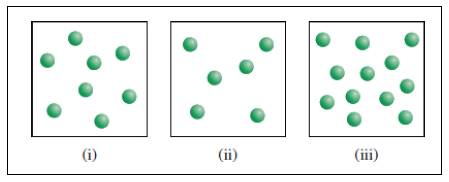
Fig (1)
The relative rates of the reaction in the given three containers can be determined as follows,
For convenience, we can use the number of molecules to represent the concentration. Therefore, the relative rates of reaction for the three containers are,
To get the relative rates dividing each rate by
Therefore
Relative rate of the reaction in three containers are
(b)
Interpretation:
To determine the relative rates be affected if the volume of the each container were doubled
Concept introduction:
Rate of the reaction is the change in the concentration of reactant or a product with time.
Rate equation for the general reaction
Explanation of Solution
The given first order reaction is,
For first order reaction,
X molecules are placed in three equal-volume containers at the same temperature.

Fig (1)
The relative rates of the reaction in the given three containers can be determined as follows,
For convenience, we can use the number of molecules to represent the concentration. Therefore, the relative rates of reaction for the three containers are,
To get the relative rates dividing each rate by
Therefore
Relative rate of the reaction in three containers are
The relative rates would be unaffected if the volume of the each container were doubled. Therefore, the relative rates between the three containers would remain same and so the actual rate would decrease by
(c)
Interpretation:
The relative half-life of the reactions in
Concept introduction:
Rate of the reaction is the change in the concentration of reactant or a product with time.
Rate equation for the general reaction
Half-life is the time required for one half of a reactant to react.
For first order reaction
Half-life for a first order reaction is
Explanation of Solution
The given first order reaction is,
For first order reaction,
X molecules are placed in three equal-volume containers at the same temperature.

Fig (1)
We know that, the half-life of a first order reaction is independent on substrate (reactant) concentration; it does not depend on substrate concentration.
Therefore, the relative half-life of the reactions in
Want to see more full solutions like this?
Chapter 13 Solutions
Chemistry
- Sucrose, a sugar, decomposes in acid solution to give glucose and fructose. The reaction is first-order in sucrose, and the rate constant at 25 C is k = 0.21 h1. If the initial concentration of sucrose is 0.010 mol/L, what is its concentration after 5.0 h?arrow_forwardThe frequency factor A is 6.31 108 L mol1 s1 and the activation energy is 10. kJ/mol for the gas-phase reaction NO(g)+O3(g)NO2(g)+O2(g) which is important in the chemistry of stratospheric ozone depletion. (a) Calculate the rate constant for this reaction at 370. K. (b) Assuming that this is an elementary reaction, calculate the rate of the reaction at 370. K if [NO] = 0.0010 M and [O3] = 0.00050 M.arrow_forwardThe hydrolysis of the sugar sucrose to the sugars glucose and fructose, C12H22O11+H2OC6H12O6+C6H12O6 follows a first-order rate equation for the disappearance of sucrose: Rate =k[C12H22O11] (The products of the reaction, glucose and fructose, have the same molecular formulas but differ in the arrangement of the atoms in their molecules.) (a) In neutral solution, k=2.11011s1 at 27 C and 8.51011s1 at 37 C. Determine the activation energy, the frequency factor, and the rate constant for this equation at 47 C (assuming the kinetics remain consistent with the Arrhenius equation at this temperature). (b) When a solution of sucrose with an initial concentration of 0.150 M reaches equilibrium, the concentration of sucrose is 1.65107M . How long will it take the solution to reach equilibrium at 27 C in the absence of a catalyst? Because the concentration of sucrose at equilibrium is so low, assume that the reaction is irreversible. (c) Why does assuming that the reaction is irreversible simplify the calculation in pan (b)?arrow_forward
- At 573 K, gaseous NO2(g) decomposes, forming NO(g) and O2(g). If a vessel containing NO2(g) has an initial concentration of 1.9 102 mol/L, how long will it take for 75% of the NO2(g) to decompose? The decomposition of NO2(g) is second-order in the reactant and the rate constant for this reaction, at 573 K, is 1.1 L/mol s.arrow_forwardFor a reaction involving the decomposition of Z at a certain temperature, the following data are obtained: (a) What is the order of the reaction? (b) Write the rate expression for the decomposition of Z. (c) Calculate k for the decomposition at that temperature.arrow_forwardFor a reaction involving the decomposition of a hypothetical substance Y, these data are obtained: Determine the order of the reaction. Write the rate law for the decomposition of Y. Calculate k for the experiment above.arrow_forward
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax
Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning





