
Chemistry
9th Edition
ISBN: 9781133611097
Author: Steven S. Zumdahl
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 13, Problem 2ALQ
The boxes shown below represent a set of initial conditions for the reaction:
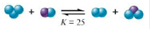
Draw a quantitative molecular picture that shows what this system looks like after the reactants are mixed in one of the boxes and the system reaches equilibrium. Support your answer with calculations.
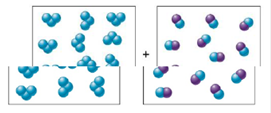
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Chapter 13 Solutions
Chemistry
Ch. 13 - Characterize a system at chemical equilibrium with...Ch. 13 - What is the law of mass action? Is it true that...Ch. 13 - Consider the following reactions at some...Ch. 13 - What is the difference between K and Kp? When doc...Ch. 13 - What are homogeneous equilibria? Heterogeneous...Ch. 13 - Distinguish between the terms equilibrium constant...Ch. 13 - Summarize the steps for solving equilibrium...Ch. 13 - A common type of reaction we will study is that...Ch. 13 - What is Le Chteliers principle? Consider the...Ch. 13 - The only stress (change) that also changes the...
Ch. 13 - Consider an equilibrium mixture of four chemicals...Ch. 13 - The boxes shown below represent a set of initial...Ch. 13 - For the reactionH2(g)+I2(g)2HI(g), consider two...Ch. 13 - Given the reactionA(g)+B(g)C(g)+D(g), consider the...Ch. 13 - Consider the reaction A(g)+2B(g)C(g)+D(g) in a...Ch. 13 - Consider the reactionA(g)+B(g)C(g)+D(g). A friend...Ch. 13 - Consider the following statements: Consider the...Ch. 13 - Le Chteliers principle is stated (Section 12-7) as...Ch. 13 - The value of the equilibrium constant K depends on...Ch. 13 - Consider an initial mixture of N2 and H2 gases...Ch. 13 - Consider the following reaction:...Ch. 13 - Consider the same reaction as in Question 11. In...Ch. 13 - Suppose a reaction has the equilibrium constant K...Ch. 13 - Suppose a reaction has the equilibrium constant K...Ch. 13 - Consider the following reaction at some...Ch. 13 - Consider the following generic reaction:...Ch. 13 - Explain the difference between K, Kp, and Q.Ch. 13 - Consider the following reactions:...Ch. 13 - For a typical equilibrium problem, the value of K...Ch. 13 - Which of the following statements is(are) true?...Ch. 13 - Write the equilibrium expression (K) for each of...Ch. 13 - Write the equilibrium expression (Kp) for each...Ch. 13 - At a given temperature, K = 1.3 102 for the...Ch. 13 - For the reaction H2(g)+Br2(g)2HBr(g) Kp = 3.5 104...Ch. 13 - For the reaction 2NO(g)+2H2(g)N2(g)+2H2O(g) it is...Ch. 13 - At high temperatures, elemental nitrogen and...Ch. 13 - At a particular temperature, a 3.0-L flask...Ch. 13 - At a particular temperature a 2.00-L flask at...Ch. 13 - The following equilibrium pressures at a certain...Ch. 13 - The following equilibrium pressures were observed...Ch. 13 - At 327c, the equilibrium concentrations are...Ch. 13 - At 1100 K, Kp = 0.25 for the reaction...Ch. 13 - Write expressions for K and Kp for the following...Ch. 13 - Write expressions for Kp for the following...Ch. 13 - For which reactions in Exercise 33 is Kp equal to...Ch. 13 - For which reactions in Exercise 34 is Kp equal to...Ch. 13 - Consider the following reaction at a certain...Ch. 13 - In a study of the reaction...Ch. 13 - The equilibrium constant is 0.0900 at 25C for the...Ch. 13 - The equilibrium constant is 0.0900 at 25C for the...Ch. 13 - Ethyl acetate is synthesized in a nonreacting...Ch. 13 - For the reaction 2H2O(g)2H2(g)+O2(g) K = 2.4 103...Ch. 13 - The reaction 2NO(g)+Br2(g)2NOBr(g) has Kp = 109 at...Ch. 13 - A 1.00-L flask was filled with 2.00 moles of...Ch. 13 - A sample of S8(g) is placed in an otherwise empty...Ch. 13 - At a particular temperature, 12.0 moles of SO3 is...Ch. 13 - At a particular temperature, 8.0 moles of NO2 is...Ch. 13 - An initial mixture of nitrogen gas and hydrogen...Ch. 13 - Nitrogen gas (N2) reacts with hydrogen gas (H2) to...Ch. 13 - At a particular temperature, K = 3.75 for the...Ch. 13 - At 2200C, Kp = 0.050 for the reaction...Ch. 13 - At 25c, K = 0.090 for the reaction...Ch. 13 - At 1100 K, KP = 0.25 for the reaction...Ch. 13 - At a particular temperature, Kp = 0.25 for the...Ch. 13 - At 35C, K = 1.6 105 for the reaction...Ch. 13 - At a particular temperature, K = 4.0 107 for the...Ch. 13 - At a particular temperature, K = 2.0 106 for the...Ch. 13 - Lexan is a plastic used to make compact discs,...Ch. 13 - Prob. 61ECh. 13 - Prob. 62ECh. 13 - Suppose the reaction system...Ch. 13 - Predict the shift in the equilibrium position that...Ch. 13 - An important reaction in the commercial production...Ch. 13 - What will happen to the number of moles of SO3 in...Ch. 13 - In which direction will the position of the...Ch. 13 - Hydrogen for use in ammonia production is produced...Ch. 13 - Old-fashioned smelling salts consist of ammonium...Ch. 13 - Ammonia is produced by the Haber process, in which...Ch. 13 - Prob. 71AECh. 13 - Given the following equilibrium constants at...Ch. 13 - Consider the decomposition of the compound C5H6O3...Ch. 13 - At 25C. Kp 1 1031 for the reaction a. Calculate...Ch. 13 - The gas arsine, AsH3, decomposes as follows:...Ch. 13 - At a certain temperature, K = 9.1 10-4 for the...Ch. 13 - At a certain temperature, K = 1.1 l03 for the...Ch. 13 - For the reaction PCl5(g)PCl3(g)+Cl2(g) at 600. K,...Ch. 13 - At 25C, gaseous SO2Cl2 decomposes to SO2(g) and...Ch. 13 - For the following reaction at a certain...Ch. 13 - Novelty devices for predicting rain contain...Ch. 13 - Consider the reaction Fe3+(aq)+SCN(aq)FeSCN2+(aq)...Ch. 13 - Chromium(VI) forms two different oxyanions, the...Ch. 13 - Prob. 84AECh. 13 - Suppose K = 4.5 103 at a certain temperature for...Ch. 13 - For the reaction below, Kp = 1.16 at 800C....Ch. 13 - Many sugars undergo a process called mutarotation,...Ch. 13 - Peptide decomposition is one of the key processes...Ch. 13 - Prob. 89AECh. 13 - Methanol, a common laboratory solvent, poses a...Ch. 13 - An equilibrium mixture contains 0.60 g solid...Ch. 13 - At a particular temperature, 8.1 moles of NO2 gas...Ch. 13 - A sample of solid ammonium chloride was placed in...Ch. 13 - In a given experiment, 5.2 moles of pure NOCl was...Ch. 13 - For the reactionN2O4(g)2NO2(g),Kp=0.25 at a...Ch. 13 - Consider the following exothermic reaction at...Ch. 13 - For the following endothermic reaction at...Ch. 13 - A 1.604-g sample of methane (CH4) gas and 6.400 g...Ch. 13 - A 4.72-g sample of methanol (CH3OH) was placed in...Ch. 13 - At 35C, K = 1.6 105 for the reaction...Ch. 13 - Nitric oxide and bromine at initial partial...Ch. 13 - At 25C. Kp = 5.3 105 for the reaction...Ch. 13 - Consider the reaction P4(g)2P2(g) where Kp = 1.00 ...Ch. 13 - The partial pressures of an equilibrium mixture of...Ch. 13 - At 125C, KP = 0.25 for the reaction...Ch. 13 - A mixture of N2, H2, and NH3 is at equilibrium...Ch. 13 - Consider the decomposition equilibrium for...Ch. 13 - An 8.00-g sample of SO3 was placed in an evacuated...Ch. 13 - A sample of iron(II) sulfate was heated in an...Ch. 13 - Prob. 111CPCh. 13 - A sample of N2O4(g) is placed in an empty cylinder...Ch. 13 - A sample of gaseous nitrosyl bromide (NOBr) was...Ch. 13 - The equilibrium constant Kp for the reaction...Ch. 13 - For the reaction NH3(g)+H2S(g)NH4HS(s) K = 400. at...Ch. 13 - Given K = 3.50 at 45C for the reaction...Ch. 13 - In a solution with carbon tetrachloride as the...Ch. 13 - The hydrocarbon naphthalene was frequently used in...Ch. 13 - A gaseous material XY(g) dissociates to some...
Additional Science Textbook Solutions
Find more solutions based on key concepts
The chapter sections to review are shown in parentheses at the end of each problem. A "chemical-free” shampoo i...
Basic Chemistry
Label each statement about the polynucleotide ATGGCG as true or false. The polynucleotide has six nucleotides. ...
General, Organic, & Biological Chemistry
1. What did each of the following scientists contribute to our knowledge of the atom?
a. William Crookes
b. E...
Chemistry For Changing Times (14th Edition)
Calculate the lattice energy of CaCl2 using a Born-Haber cycle and data from Appendices F and L and Table 7.5. ...
Chemistry & Chemical Reactivity
Q1. What is the empirical formula of a compound with the molecular formula
Chemistry: A Molecular Approach
45. Calculate the mass of nitrogen dissolved at room temperature in an 80.0-L home aquarium. Assume a total pre...
Chemistry: Structure and Properties
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- The boxes shown below represent a set of initial conditions for the reaction: Draw a quantitative molecular picture that shows what this system looks like after the reactants are mixed in one of the boxes and the system reaches equilibrium. Support your answer with calculations. Consider an equilibrium mixture of four chemicals (A, B, C, and D, all gases) reacting in a closed flask according to the foll owing equation: A+BC+D a. You add more A to the flask. How does the concentration of each chemical compare to its original concentration after equilibrium is re-established? Justify your answer. b. You have the original set-up at equilibrium, and add more D to the flask. How does the concentration of each chemical compare to its original concentration after equilibrium is re-established? Justify your answer.arrow_forward. Explain what it means that a reaction has reached a state of chemical equilibrium. Explain why equilibrium is a dynamic state: Does a reaction really “stop” when the system reaches a state of equilibrium? Explain why, once a chemical system has reached equilibrium, the concentrations of all reactants remain constant with time. Why does this constancy of concentration not contradict our picture of equilibrium as being dynamic? What happens to the rates of the forward and reverse reactions as a system proceeds to equilibrium from a starting point where only reactants are present?arrow_forwardHow does equilibrium represent the balancing of opposing processes? Give an example of an “equilibrium” encountered in everyday life, showing how the processes involved oppose each other.arrow_forward
- Explain that equilibrium is dynamic, and that at equilibrium the forward and backward reaction rates are equal.arrow_forwardTell what will happen to each equilibrium concentration in the following when the indicated stress is applied and a new equilibrium position is established. a. LiOH(s)+CO2(g)LiHCO3(s)+heat; CO2 is removed. b. 2NaHCO3(s)+heatNa2O(s)+2CO2(g)+H2O(g); The system is cooled. c. CaCO3(s)+heatCaO(s)+CO2(g); The system is cooled.arrow_forwardEqual amounts of two gases, A and B3, are placed in an evacuated container and react to form AB and B2. A(g)+B3(g)AB(g)+B2(g) The molecular scale diagram represents the relative amounts of each substance present once equilibrium is reached. Which of the following graphs best describes the progress of the reaction as it proceeds to equilibrium? Explain why the graph you chose must be the correct one.arrow_forward
- For the reaction C6H6(g)+3H2(g)C6H12(g)+heat determine in what direction the equilibrium will be shifted by each of the following changes. Decreasing the concentration of H2 a. Increasing the concentration of C6H6 b. Decreasing the temperature c. Increasing the pressure by decreasing the volume of the containerarrow_forward. For the reaction system C(s)+H2O(g)H2(g)+CO(g)which has already reached a state of equilibrium, predict the effect that each of the following changes will have on the position of the equilibrium. Tell whether the equilibrium will shift to the right, will shift to the left, or will not be affected. a. The pressure of hydrogen is increased by injecting an additional mole of hydrogen gas into the reaction vessel. b. Carbon monoxide gas is removed as it forms by use of a chemical absorbent or “scrubber.” c. An additional amount of solid carbon is added to the reaction vessel.arrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you
 World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
 Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning

World of Chemistry, 3rd edition
Chemistry
ISBN:9781133109655
Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:Brooks / Cole / Cengage Learning

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:9781305079243
Author:Steven S. Zumdahl, Susan A. Zumdahl
Publisher:Cengage Learning


Introductory Chemistry: A Foundation
Chemistry
ISBN:9781337399425
Author:Steven S. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

General, Organic, and Biological Chemistry
Chemistry
ISBN:9781285853918
Author:H. Stephen Stoker
Publisher:Cengage Learning
Chemical Equilibria and Reaction Quotients; Author: Professor Dave Explains;https://www.youtube.com/watch?v=1GiZzCzmO5Q;License: Standard YouTube License, CC-BY