
Organic Chemistry
9th Edition
ISBN: 9781305080485
Author: John E. McMurry
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Question
Chapter 13.11, Problem 19P
Interpretation Introduction
Interpretation:
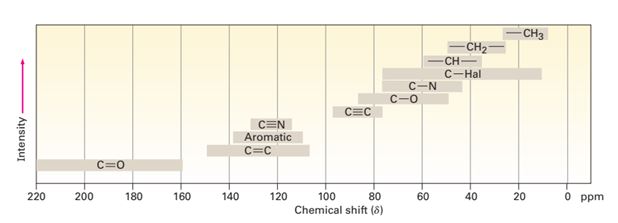
Most 13C resonances are between 0 and 220 ppm downfield from the TMS reference line, with the exact chemical shift of each 13C resonance dependent on that carbon’s electronic environment within the molecule.
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
Analyse the 13C NMR spectrum for 1,3-dibromobenzene
How could 13C NMR spectroscopy be used to distinguish among isomers X, Y, and Z?
11-Explain why 2-chloropropene shows signals for three kinds of protons in its 1H NMRspectrum? Draw the structure and predict the approximate values for 1H-NMR spectra.
Chapter 13 Solutions
Organic Chemistry
Ch. 13.1 - Prob. 1PCh. 13.1 - Prob. 2PCh. 13.2 - Prob. 3PCh. 13.3 - The following 1H NMR peaks were recorded on a...Ch. 13.3 - When the 1Η NMR spectrum of acetone, CH3COCH3, is...Ch. 13.4 - Each of the following compounds has a single 1H...Ch. 13.4 - Identify the different types of protons in the...Ch. 13.5 - How many peaks would you expect in the 1H NMR...Ch. 13.6 - Predict the splitting patterns you would expect...Ch. 13.6 - Draw structures for compounds that meet the...
Ch. 13.6 - The integrated 1H NMR spectrum of a compound of...Ch. 13.7 - Identify the indicated sets of protons as...Ch. 13.7 - How many kinds of electronically nonequivalent...Ch. 13.7 - How many absorptions would you expect (S)-malate,...Ch. 13.8 - 3-Bromo-1-phenyl-1-propene shows a complex NMR...Ch. 13.9 - How could you use 1H NMR to determine the...Ch. 13.11 - Prob. 17PCh. 13.11 - Propose structures for compounds that fit the...Ch. 13.11 - Prob. 19PCh. 13.12 - Prob. 20PCh. 13.12 - Prob. 21PCh. 13.12 - Prob. 22PCh. 13.13 - Prob. 23PCh. 13.SE - Into how many peaks would you expect the 1H NMR...Ch. 13.SE - How many absorptions would you expect the...Ch. 13.SE - Sketch what you might expect the 1H and 13C NMR...Ch. 13.SE - How many electronically nonequivalent kinds of...Ch. 13.SE - Identify the indicated protons in the following...Ch. 13.SE - Prob. 29APCh. 13.SE - Prob. 30APCh. 13.SE - When measured on a spectrometer operating at 200...Ch. 13.SE - Prob. 32APCh. 13.SE - Prob. 33APCh. 13.SE - How many types of nonequivalent protons are...Ch. 13.SE - The following compounds all show a single line in...Ch. 13.SE - Prob. 36APCh. 13.SE - Propose structures for compounds with the...Ch. 13.SE - Predict the splitting pattern for each kind of...Ch. 13.SE - Predict the splitting pattern for each kind of...Ch. 13.SE - Identify the indicated sets of protons as...Ch. 13.SE - Identify the indicated sets of protons as...Ch. 13.SE - The acid-catalyzed dehydration of...Ch. 13.SE - How could you use 1H NMR to distinguish between...Ch. 13.SE - Propose structures for compounds that fit the...Ch. 13.SE - Propose structures for the two compounds whose 1H...Ch. 13.SE - Prob. 46APCh. 13.SE - How many absorptions would you expect to observe...Ch. 13.SE - Prob. 48APCh. 13.SE - How could you use 1H and 13C NMR to help...Ch. 13.SE - How could you use 1H NMR, 13C NMR, and IR...Ch. 13.SE - Assign as many resonances as you can to specific...Ch. 13.SE - Assume that you have a compound with the formula...Ch. 13.SE - The compound whose 1H NMR spectrum is shown has...Ch. 13.SE - The compound whose 1H NMR spectrum is shown has...Ch. 13.SE - Propose structures for compounds that fit the...Ch. 13.SE - Long-range coupling between protons more than two...Ch. 13.SE - The 1H and 13C NMR spectra of compound A, C8H9Br,...Ch. 13.SE - Propose structures for the three compounds whose...Ch. 13.SE - The mass spectrum and 13C NMR spectrum of a...Ch. 13.SE - Compound A, a hydrocarbon with M+=96 in its mass...Ch. 13.SE - Propose a structure for compound C, which has...Ch. 13.SE - Prob. 62GPCh. 13.SE - Propose a structure for compound E, C7H12O2, which...Ch. 13.SE - Compound F, a hydrocarbon with M+=96 in its mass...Ch. 13.SE - 3-Methyl-2-butanol has five signals in its 13C NMR...Ch. 13.SE - A 13C NMR spectrum of commercially available...Ch. 13.SE - Carboxylic acids (RCO2H) react with alcohols (ROH)...Ch. 13.SE - Prob. 68GPCh. 13.SE - The proton NMR spectrum is shown for a compound...Ch. 13.SE - The proton NMR spectrum of a compound with the...Ch. 13.SE - The proton NMR spectrum is shown for a compound...
Knowledge Booster
Similar questions
- can you list the corresponding ppm values for the non-aromatic carbon atom in ^13C NMR spectrum for benzonitrile?arrow_forwardWhat characteristics of the methyl benzoate spectrum rule out an aldehyde or carboxylicacid functional group giving the absorption at 1723 cm-1?arrow_forwardWhat does the H-NMR peaks say about the C9H12 structure?arrow_forward
- Fully interpret and compare the 13C NMR spectrum and the 1H NMR spectrum of the compound (R,R)-N,N’-bis(3,5-di-tert-butylsalicylidene)-1,2-cyclohexanediamine. Label and explain the peaks and why they show up where they do.arrow_forwardHow many signals are possible in the 13C NMR Spectrum of 1-butene? Please provide the structure and signals relevant to the atomsarrow_forwardSketch the spectra that would be obtained for 2-chloroethanol: The 13C NMR spectrum.arrow_forward
- Assign the chemical shifts δ 1.1, δ 1.7, δ 2.0, and δ 2.3 to the appropriate protons of 2-pentanone.arrow_forwardInterpret the main features (not details) of the 1H NMR spectrum of coumarin (2), Spec 2. In particular, deduce the value of the coupling constant (J) between the two alkene protons.arrow_forwardConsider the aromatic compound 4-isopropyl-benzonitrile. (Benzonitrile is a benzene ring with the nitrile group on position 1.) How many signals for non-equivalent types of protons will be in its proton NMR spectrum?arrow_forward
- How many signals would you expect to observe in the 1H -NMR spectrum of 1,3-dichloropropane? What would be the multiplicity (splitting) of each signal?arrow_forwardPlease describe the key characteristics of the H-NMR and 13C-NMR spectra.arrow_forwardMay I please get some help with 13C NMR for C6H10O I need help with tabulating chemical shifts, splitting, neighboring protons, integration, fragments. Are there any special features?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

 Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole
Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole


Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Macroscale and Microscale Organic Experiments
Chemistry
ISBN:9781305577190
Author:Kenneth L. Williamson, Katherine M. Masters
Publisher:Brooks Cole