
Concept explainers
Interpretation:
The structure of compound of given molecular formula C7H12O2 to be predicted using 13CNMR spectra.
Concept introduction:
The 13CNMR spectrum gives information on the different electronic environments of carbon. As like 1HNMR, the number of signals generated in 13CNMR are predicted by performing symmetry operations (rotation or reflection symmetry). Only chemical shift values are reported in the spectrum but not the multiplicity and integration values because the coupling between two neighboring 13C-13 C nuclei are weakly involved due to the low abundance of 13C isotopes of carbon atom.
To Identify:
The structure of given molecular formula C7H12O2.
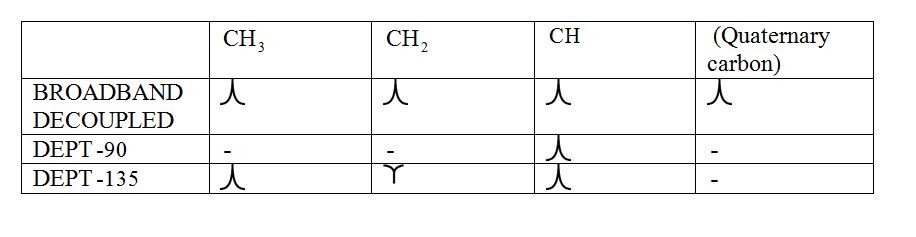
Broadband-decoupled 13CNMR spectrum:
The spectra provide information regarding the total number of carbon environments.
DEPT (Distortionless enhancement by polarization transfer):
a) DEPT-90: The spectrum exhibits signal only from CH group and no signals from CH3, CH2, CH and quaternary carbon (carbon with no protons).
b) DEPT-135: The spectrum exhibits CH3 groups and CH groups as positive signals (pointing up); CH2 groups appear as negative signals (pointing down) and quaternary carbon does not appear.
The signals appear in each type of spectrum:
Want to see the full answer?
Check out a sample textbook solution
Chapter 13 Solutions
Organic Chemistry
- Following are two constitutional isomers with the molecular formula C4H8O2. (a) Predict the number of signals in the 1H-NMR spectrum of each isomer. (b) Predict the ratio of areas of the signals in each spectrum. (c) Show how you can distinguish between these isomers on the basis of chemical shift.arrow_forwardFollowing is the mass spectrum of an unknown compound. The two highest peaks are at m/z 120 and 122. Suggest a structure for this compound. (Data from http://webbook.nist.gov/chemistry/.)arrow_forwardAssign each peak on the following spectrum.arrow_forward
- 1. The given spectra are for a compound with the formula C9H12O. a. Using the provided shift options, assign the correct shifts to the indicated regions in the 1H NMR spec, you will not use all. b. Using the 1H NMR spec you just labeled along with the provided IR spec, determine and draw the structure of the compound that these spectra belong to.arrow_forwardIdentify the important peaks in the following MS spectral data and draw the structure of the important peaks in the following MS spectral data.arrow_forwardThe methyl protons in acetaldehyde appear at a shift of 2.20 ppm, while the aldehydic proton appears at 9.80 ppm. In a 60 MHz NMR with an external field of 1.5 T, what is the magnetic field experienced by each type of proton? Explain why the presence of 3 protons on the adjacent carbon split the aldehydic proton peak into a quartet.arrow_forward
- A signal is seen at 600 Hz from the TMS signal in an NMR spectrometer with a 300-MHz operating frequency. a. What is the chemical shift of the signal? b. What is its chemical shift in an instrument operating at 500 MHz? c. How many hertz from the TMS signal is the signal in a 500-MHz spectrometer?arrow_forwardIdentify the solvent peak in the spectrum and list its chemical shift. Based on the chemical shifts, what functional groups are present in your compound? For each, correlate the functional group with the chemical shift of the identifying peak. List all the 13C NMR data in ACS journal style. The format is: 13C NMR (125 MHz, CDCl3): d list chemical shifts here.arrow_forwardA carbon signal measured in a 300MHz spectrometer with an observing frequency of 75.47MHz isfound to have a chemical shift of 187.23 ppm. What is the resonance frequency of the carbon withthis chemical shift?A) 56169 HzB) 75 484 130 HzC) 14 130 HzD) 300 056 139 Hzarrow_forward

 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole
Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole



