
Chemistry & Chemical Reactivity
10th Edition
ISBN: 9781337399074
Author: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 13.2, Problem 13.2CYU
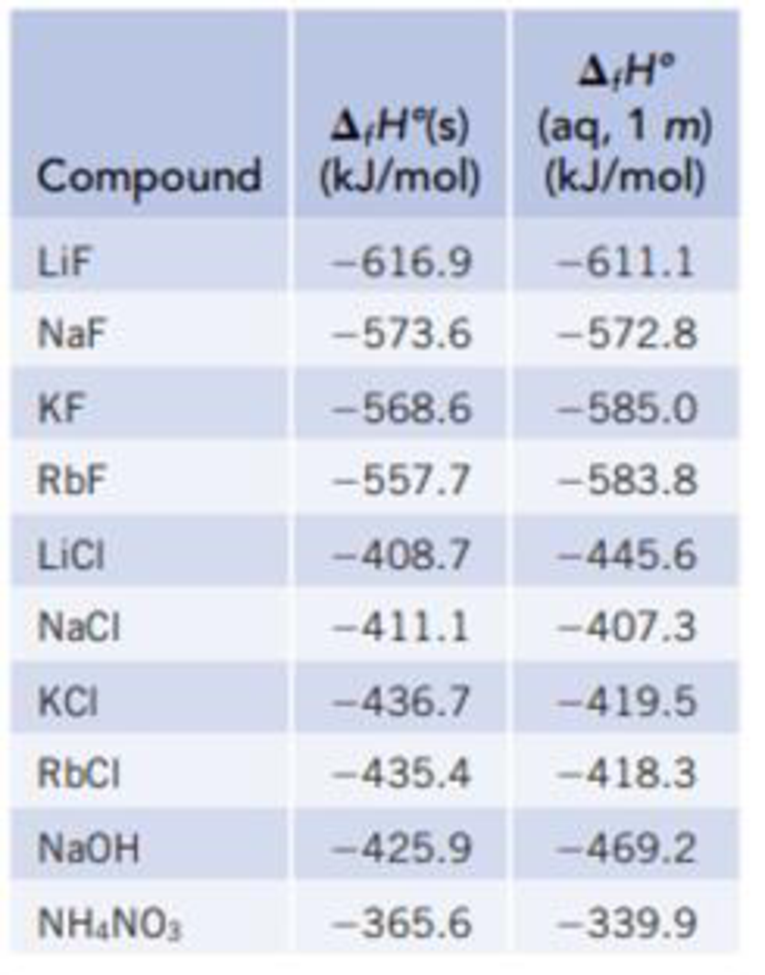
Use the data in Table 13.1 to calculate the enthalpy of solution for NaOH.
TABLE 13.1
Data for calculating enthalpy of Solution
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
Calculate the freezing point of a water solution at each concentration.
1. 0.85 m
2. 1.35 m
3. 5.2 m
4. 2.35 m
Calculate the freezing point of a 0,10 m aqueous solution of AlCl
Phosphoric acid is usually obtained as an 85% phosphoric acid solution. If it is15M.Calculate:a. Density of the solution b. Molality c. NormalityAtomic Weights, g/mol: O =16 Na = 23 H=1 P= 31
Chapter 13 Solutions
Chemistry & Chemical Reactivity
Ch. 13.1 - (a) If you dissolve 10.0 g (about one heaping...Ch. 13.2 - Use the data in Table 13.1 to calculate the...Ch. 13.3 - Prob. 13.3CYUCh. 13.4 - Assume you dissolve 10.0 g of sucrose (C12H22O11)...Ch. 13.4 - What quantity of ethylene glycol, HOCH2CH2OH, must...Ch. 13.4 - In the northern United States, summer cottages are...Ch. 13.4 - Bradykinin is a small peptide (9 amino acids; 1060...Ch. 13.4 - An aluminum-containing compound has the empirical...Ch. 13.4 - A 1.40-g sample of polyethylene, a common plastic,...Ch. 13.4 - Calculate the freezing point of 525 g of water...
Ch. 13.5 - The blue line on the diagram illustrates the...Ch. 13.5 - How many theoretical plates are required to...Ch. 13.5 - Prob. 1.3ACPCh. 13.5 - The vapor pressure of pure heptane is 361.5 mm Hg...Ch. 13.5 - If the headspace of a soda is 25 mL and the...Ch. 13.5 - Prob. 2.2ACPCh. 13.5 - Prob. 2.3ACPCh. 13.5 - Prob. 2.4ACPCh. 13.5 - Prob. 3.1ACPCh. 13.5 - Prob. 3.2ACPCh. 13.5 - Prob. 3.3ACPCh. 13 - You dissolve 2.56 g of succinic acid, C2H4(CO2H)2,...Ch. 13 - You dissolve 45.0 g of camphor, C10H16O, in 425 mL...Ch. 13 - Prob. 3PSCh. 13 - Prob. 4PSCh. 13 - Prob. 5PSCh. 13 - Prob. 6PSCh. 13 - Prob. 7PSCh. 13 - Prob. 8PSCh. 13 - Hydrochloric acid is sold as a concentrated...Ch. 13 - Concentrated sulfuric acid has a density of 1.84...Ch. 13 - The average lithium ion concentration in seawater...Ch. 13 - Silver ion has an average concentration of 28 ppb...Ch. 13 - Which pairs of liquids will be miscible? (a) H2O...Ch. 13 - Acetone, CH3COCH3, is quite soluble in water....Ch. 13 - Prob. 15PSCh. 13 - Use the following data to calculate the enthalpy...Ch. 13 - You make a saturated solution of NaCl at 25 C. No...Ch. 13 - Some lithium chloride, LiCl, is dissolved in 100...Ch. 13 - Prob. 19PSCh. 13 - The Henrys law constant for O2 in water at 25 is...Ch. 13 - An unopened soda can has an aqueous CO2...Ch. 13 - Hydrogen gas has a Henrys law constant of 7.8 104...Ch. 13 - A sealed flask contains water and oxygen gas at 25...Ch. 13 - Butane, C4H10, has been suggested as the...Ch. 13 - A 35.0-g sample of ethylene glycol, HOCH2CH2OH, is...Ch. 13 - Urea, (NH2)2CO, which is widely used in...Ch. 13 - Pure ethylene glycol, HOCH2CH2OH, is added 2.00 kg...Ch. 13 - Pure iodine (105 g) is dissolved in 325 g of CCl4...Ch. 13 - Prob. 29PSCh. 13 - What is the boiling point of a solution composed...Ch. 13 - Prob. 31PSCh. 13 - Prob. 32PSCh. 13 - Prob. 33PSCh. 13 - Some ethylene glycol, HOCH2CH2OH, is added to your...Ch. 13 - You dissolve 15.0 g of sucrose, C12H22O11, in a...Ch. 13 - A typical bottle of wine consists of an 11%...Ch. 13 - Prob. 37PSCh. 13 - Estimate the osmotic pressure of human blood at 37...Ch. 13 - An aqueous solution containing 1.00 g of bovine...Ch. 13 - Calculate the osmotic pressure of a 0.0120 M...Ch. 13 - You add 0.255 g of an orange, crystalline compound...Ch. 13 - Butylated hydroxyanisole (BHA) is used in...Ch. 13 - Benzyl acetate is one of the active components of...Ch. 13 - Anthracene, a hydrocarbon obtained from coal, has...Ch. 13 - An aqueous solution contains 0.180 g of an...Ch. 13 - Aluminon, an organic compound, is used as a...Ch. 13 - Prob. 47PSCh. 13 - To make homemade ice cream, you cool the milk and...Ch. 13 - List the following aqueous solutions in order of...Ch. 13 - Arrange the following aqueous solutions in order...Ch. 13 - When solutions of BaCl2 and Na2SO4 are mixed, the...Ch. 13 - The dispersed phase of a certain colloidal...Ch. 13 - Phenylcarbinol is used in nasal sprays as a...Ch. 13 - (a) Which aqueous solution is expected to have the...Ch. 13 - Arrange the following aqueous solutions in order...Ch. 13 - Prob. 56GQCh. 13 - Dimethylglyoxime [DMG, (CH3CNOH)2] is used as a...Ch. 13 - A 10.7 m solution of NaOH has a density of 1.33...Ch. 13 - Concentrated aqueous ammonia has a molarity of...Ch. 13 - Prob. 60GQCh. 13 - If you want a solution that is 0.100 m in ions,...Ch. 13 - Consider the following aqueous solutions: (i) 0.20...Ch. 13 - (a) Which solution is expected to have the higher...Ch. 13 - The solubility of NaCl in water at 100 C is 39.1...Ch. 13 - Instead of using NaCl to melt the ice on your...Ch. 13 - The smell of ripe raspberries is due to...Ch. 13 - Hexachlorophene has been used in germicidal soap....Ch. 13 - The solubility of ammonium formate, NH4CHO2, in...Ch. 13 - How much N2 can dissolve in water at 25 C if the...Ch. 13 - Cigars are best stored in a humidor at 18 C and...Ch. 13 - An aqueous solution containing 10.0 g of starch...Ch. 13 - Prob. 72GQCh. 13 - Calculate the enthalpies of solution for Li2SO4...Ch. 13 - Water at 25 C has a density of 0.997 g/cm3....Ch. 13 - If a volatile solute is added to a volatile...Ch. 13 - A solution is made by adding 50.0 mL of ethanol...Ch. 13 - A 2.0% (by mass) aqueous solution of novocainium...Ch. 13 - A solution is 4.00% (by mass) maltose and 96.00%...Ch. 13 - The following table lists the concentrations of...Ch. 13 - A tree is 10.0 m tall. (a) What must be the total...Ch. 13 - Prob. 81GQCh. 13 - A compound is known to be a potassium halide, KX....Ch. 13 - Prob. 85GQCh. 13 - If one is very careful, it is possible to float a...Ch. 13 - A solution of benzoic acid in benzene has a...Ch. 13 - You dissolve 5.0 mg of iodine, I2, in 25 mL of...Ch. 13 - Prob. 89ILCh. 13 - In a police forensics lab, you examine a package...Ch. 13 - An organic compound contains carbon (71.17%),...Ch. 13 - Prob. 92ILCh. 13 - When sails of Mg2+, Ca2+, and Be2+ are placed in...Ch. 13 - Explain why a cucumber shrivels up when it is...Ch. 13 - Prob. 95SCQCh. 13 - A 100.-gram sample of sodium chloride (NaCl) is...Ch. 13 - Prob. 97SCQCh. 13 - Prob. 98SCQCh. 13 - Starch contains CC, CH, CO, and OH bonds....Ch. 13 - Prob. 100SCQCh. 13 - You have two aqueous solutions separated by a...Ch. 13 - Prob. 102SCQCh. 13 - Sodium chloride (NaCl) is commonly used to melt...Ch. 13 - Prob. 105SCQCh. 13 - Prob. 106SCQCh. 13 - Prob. 107SCQ
Additional Science Textbook Solutions
Find more solutions based on key concepts
Q2. Which statement best defines chemistry?
a. The science that studies solvents, drugs, and insecticides
b. Th...
Introductory Chemistry (6th Edition)
Give the IUPAC name for each compound.
Organic Chemistry
4. 38 Strontium has four naturally occurring isotopes, with mass numbers 84, 86, 87, arid 88.
a. Write the atom...
Basic Chemistry (5th Edition)
The chapter sections to review are shown in parentheses at the end of each problem. A "chemical-free” shampoo i...
Basic Chemistry
The active ingredient in Tylenol and a host of other over-the-counter pain relievers is acetaminophen (C8H9NO2)...
Chemistry: Atoms First
Classify each example of molecular art as a pure element, a pure compound, or a mixture.
General, Organic, and Biological Chemistry - 4th edition
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- The freezing point of 0.109 m aqueous formic acid is 0.210C. Formic acid, HCHO2, is partially dissociated according to the equation HCHO2(aq)H+(aq)+CHO2(aq) Calculate the percentage of HCHO2 molecules that are dissociated, assuming the equation for the freezing-point depression holds for the total concentration of molecules and ions in the solution.arrow_forwardCalculate the percent by mass of KBr in a saturated solution of KBr in water at 10 °C. See Figure 11.17 for useful data, and report the computed percentage to one significant digit.arrow_forwardA 0.109 mol/kg aqueous solution of formic acid, HCOOH, freezes at −0.210 °C. Calculate the percent dissociation of formic acid.arrow_forward
- If one is very careful, it is possible to float a needle on the surface of water. (If the needle is magnetized, it will turn to point north and south and become a makeshift compass.) What would happen to the needle if a drop of liquid soap is added to the solution? Explain the observation.arrow_forwardGive one example of each: a salt whose heat of solution is exothermic and a salt whose heat of solution is endothermic.arrow_forwardFrom the data presented in Figure 12.11, determine which has the more positive enthalpy of solution: NaCl or NH4Cl. Explain.arrow_forward
- Explain why solutions of HBr in benzene (a nonpolar solvent) are nonconductive, while solutions in water (a polar solvent) are conductive.arrow_forwardCorrelate the degree of change in freezing point of water to the number of ions formed when salts are dissolved in water.arrow_forwardWhen lithium iodide (LiI) is dissolved in water, the solution becomes hotter.a. Is the dissolution of lithium iodide endothermic or exothermic?b. What can you conclude about the relative magnitudes of the lattice energy of lithium iodide and its heat of hydration?c. Sketch a qualitative energy diagram similar to Figure 13.7 for the dissolution of LiI.d. Why does the solution form? What drives the process?arrow_forward
- The concentration of concentrated hydrochloric acid (HC1) is around 32 percent mass by volume. Calculate the values of the following concentration units of concentrated hydrochloric acid in 1 bottle (1000 ml): a.) Molarity b.) Molality Note: Assume hydrochloric acid and water are the only components of the muriatic acid. The density of hydrochloric acid and water are 1.2 g/mL and 0.997 g/mL at room temperature, respectively.arrow_forwardThe vapor pressure of pure water is lower than the vapor pressure of a 3.20 m solution.arrow_forwardCalculate the boiling point of a 0.44 m aqueous solution of AlCl3arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax
Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781337399074
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:9780078746376
Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:Glencoe/McGraw-Hill School Pub Co

General, Organic, and Biological Chemistry
Chemistry
ISBN:9781285853918
Author:H. Stephen Stoker
Publisher:Cengage Learning

General Chemistry - Standalone book (MindTap Cour...
Chemistry
ISBN:9781305580343
Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:9781938168390
Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:OpenStax

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781133949640
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning
General Chemistry | Acids & Bases; Author: Ninja Nerd;https://www.youtube.com/watch?v=AOr_5tbgfQ0;License: Standard YouTube License, CC-BY