
Concept explainers
(a)
Interpretation:
The IUPAC name for the following alkene should be determined:
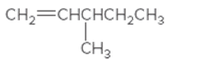
Concept Introduction:
(b)
Interpretation:
The IUPAC name for the following alkene should be determined:
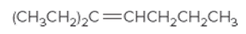
Concept Introduction:
Alkenes are named in the same way as alkanes, but the alkenes are identified by the suffix −ene, replaced instead of the ending of the name of the parent alkane. The longest carbon chain should be numbered in a way that gives the double bond the lower number. Then the compound should be named using the first number assigned to the double bond. The names of the substituents should be written first in the alphabetic order with their position on the chain.
(c)
Interpretation:
The IUPAC name for the following alkene should be determined:
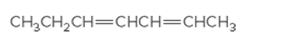
Concept Introduction:
Alkenes are named in the same way as alkanes, but the alkenes are identified by the suffix −ene, replaced instead of the ending of the name of the parent alkane. The longest carbon chain should be numbered in a way that gives the double bond the lower number. Then the compound should be named using the first number assigned to the double bond. The names of the substituents should be written first in the alphabetic order with their position on the chain.
(d)
Interpretation:
The IUPAC name for the following alkene should be determined:
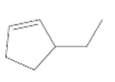
Concept Introduction:
In the nomenclature of cycloalkenes, the double bond is always located between C-1 and C-2. And 1 is not mentioned in the name. the ring is then numbered in a way that the first substituent getting the lowest number.
Want to see the full answer?
Check out a sample textbook solution
Chapter 13 Solutions
General, Organic, and Biological Chemistry - 4th edition
- Draw the following compounds: a) 6-[1-chloropropyl]-5-methyl-1-decyne b) 2-bromo-3-methyl-3,5-octadienec) 4,6,6-trimethylcyclooctened) 2-Bromobicyclo[3.3.1]nonane e) 2-Methylbicyclo[2.2.2]octane f) 1,2-Dichlorocyclohexeneg) 4,5-Dibromo-1-pentenearrow_forwardFor each molecular formula, draw all the isomeric alkynes, and give their IUPAC names.Circle the acetylenic hydrogen of each terminal alkyne.(a) C5H8 (three isomers)arrow_forward1: Draw the chain conformer of... 1a. Cyclohexane, label all the axial and equatorial hydrogens 1b. The most stable conformer of ethylcyclohexane 1c. The most sable conformer of trans-1-tert-butyl-3-methylcyclohexanearrow_forward
- CH3-CH-O+CH3MgClarrow_forwardConsider the tricyclic structure B. (a) Label each substituent on the rings as axial or equatorial. (b) Draw B using chair conformations for each six-membered ring. (c) Label the atoms on the ring fusions (the carbons that join each set of two rings together) as cis or trans to each other.arrow_forward(a) Draw all constitutional isomers formed by monochlorination of each alkane with Cl2 and hv. (b) Draw the major monobromination product formed by heating each alkane with Br2.arrow_forward
- For rotation about the C3-C4 bond of 2-chlorohexane: a) Draw a Newman projection showing the most stable conformation. b) Draw a Newman projection showing the least stable conformation.arrow_forward1. Alkene A is ___ stable than alkene B and will release ___ energy when it is hydrogenated to form 2,5-dimethylheptane.arrow_forwardThe cis ketone A is isomerized to a trans ketone B with aqueous NaOH. A similar isomerization does not occur with ketone C. (a) Draw the structure of B using a chair cyclohexane. (b) Label the substituents in C as cis or trans, and explain the difference in reactivity.arrow_forward
- Compounds E and F are two isomers of 2,3-dibromopentane drawn in staggered conformations. Which compounds (A–D) in Figure 5.8 are identical to E and F?arrow_forwardPair the following A. alkene B. alcohol C. aldehyde D. carboxylic acid E. ester F. ether G. alkyne CH3-CH2-CH2-CH=O ______ CH2-CH2-CH2-CH2-CH2-OH ______ CH3-CH2-CH2-C=O OCH3 CH3-CH2-CH2-O-CH2-CH3 CH3-CH2-CH2-C=O OH CH3-NH-CH2-CH2-CH3 ______ CH3-CH2-CH=CH-CH3 ______arrow_forwarda model of cyclohexane in a chair conformation, and explain why the names “axial” and“equatorial’ are appropriate.arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
