
Concept explainers
Interpretation:
The structure of a given molecular formula C8H16 to be predicted using spectrum details.
Concept introduction:
1HNMR : The 1HNMR spectrum gives information on the different electronic environment of protons. The number of signal (proton types) generated in 1HNMR are predicted by performing symmetry operations (rotation or reflection symmetry).
The 13CNMR spectrum gives information on the different electronic environments of carbon. As like 1HNMR, the number of signals generated in 13CNMR are predicted by performing symmetry operations (rotation or reflection symmetry). Only chemical shift values are reported in the spectrum but not the multiplicity and integration values because the coupling between two neighboring 13C - 13C nuclei are weakly involved due to the low abundance of 13C isotopes of carbon atom.
HDI Calculation:
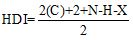
Where
C represent number of carbons.
N represent number of nitrogens.
H represent number of hydrogens.
X represent number of halogens.
To identify:
The structure of a given molecular formula C8H16.
Trending nowThis is a popular solution!

Chapter 13 Solutions
Organic Chemistry
- The following 1H NMR peaks were recorded on a spectrometer operating at 200 MHz. Convert each into δ units. (a) CHCl3; 1454 Hz (b) CH3Cl; 610 Hz (c) CH3OH; 693 Hz (d) CH2Cl2; 1060 Hzarrow_forward3-Bromo-1-phenyl-1-propene shows a complex NMR spectrum in which the vinylic proton at C2 is coupled with both the C1 vinylic proton (J = 16 Hz) and the C3 methylene protons (J = 8 Hz). Draw a tree diagram for the C2 proton signal, and account for the fact that a five-line multiplet is observed.arrow_forwardWhen the 1HNMR spectrum of an alcohol is run in dimethylsulfoxide (DMSO) solvent rather than in chloroform, exchange of the Ο-H proton is slow and spin-spin splitting is seen between the Ο-H proton and C-H protons on the adjacent carbon. What spin multiplicities would you expect for the hydroxyl protons in the following alcohols? (a) 2-Methyl-2-propanol (b) Cyclohexanol (c) Ethanol (d) 2-Propanol (e) Cholesterol (f) 1-Methylcyclohexanolarrow_forward
- What characteristics of the methyl benzoate spectrum rule out an aldehyde or carboxylicacid functional group giving the absorption at 1723 cm-1?arrow_forwardHow many signals would you expect to observe in the 1H -NMR spectrum of 2,3-dimethyl-1,3-cyclopentadiene? What would be the multiplicity (splitting) of each signal?arrow_forwardThe 13C-NMR spectrum of 3-methyl-2-butanol shows signals at d 17.88 (CH3), 18.16 (CH3), 20.01 (CH3), 35.04 (carbon-3), and 72.75 (carbon-2). Account for the fact that each methyl group in this molecule gives a different signal.arrow_forward
- Label the 13C spectrum of (R,R)-N,N’-bis(3,5-di-tert-butylsalicylidene)-1,2-cyclohexanediamine peaks and explain why they appear at the ppm that they show up at.arrow_forwardWith 1H-NMR, how many signals/peaks would you expect for 1-chloro-3-methylbutane?arrow_forwardHow many peaks (13C) would be evident in the decoupled spectrum of a. methylcyclohexane b. cyclohexene c. 1-methylcyclohexene *show structure in the solutionarrow_forward
- The intensities of peaks at 69 m/z, 70 m/z, and 71 m/z are 1.2, 31.2, and 1.7, respectively. How many carbons does the compound have?arrow_forwardThe 1H NMR spectrum of chloromethane recorded on a 300 Hz NMR spectrometer consists of signals at 444 Hz and 1071 Hz downfield from TMS. Calculate the chemical shift of each absorption. Give answer in parts per million.arrow_forwardHow many signals in 13C-NMR and 1H-NMR will be obtained from 5-methylidenocyclopenta-1,3-diene in its most stable conformation?arrow_forward
 Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
 Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole
Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning



