
Concept explainers
In the following compound, identify whether each lone pair is available to function as a base, and explain your choice:
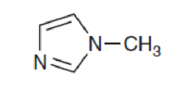
Interpretation:
The availability of lone pair in the compound to function as a base has to be explained.
Concept Introduction:
If a lone pair is not a part of the ring means not taking part in resonance, then it will be available to function as a base. It also can be explained in other way. Like if a lone pair is present in a
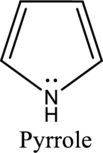
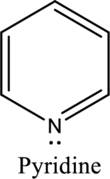
Nitrogen in both pyrrole and pyridine has a lone pair and it is
Explanation of Solution
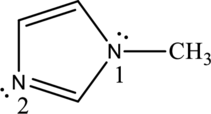
By properly examining the structure, it can be seen that the numbered
The numbered
Want to see more full solutions like this?
Chapter 1 Solutions
Organic Chemistry As a Second Language: Second Semester Topics
Additional Science Textbook Solutions
Inorganic Chemistry
General, Organic, and Biological Chemistry (3rd Edition)
Living by Chemistry
Living By Chemistry: First Edition Textbook
Elementary Principles of Chemical Processes, Binder Ready Version
- Complete the equation for the reaction between each Lewis acid-base pair. In each equation, label which starting material is the Lewis acid and which is the Lewis base; use curved arrows to show the flow of electrons in each reaction. In doing this problem, it is essential that you show valence electrons for all atoms participating in each reaction. (a) (b) (c) (d)arrow_forwardFor each molecule below, draw the conjugate acid or conjugate base or both if the molecule hasboth a conjugate acid and a conjugate base (e.g., water).arrow_forwardThiocyanic acid (HSCN) is an inorganic acid (pKa = 1.1 at 25 °C) that can be classified as “strong” largely due to resonance stabilization of its conjugate base, thiocyanate (SCN– ). In the space provided below, draw Lewis electron dot structures of SCN- and all of its important resonance forms.arrow_forward
- Complete each acid-base reaction and predict whether the position of equilibrium lies toward the left or toward the right. (a) CH3CCH+CH3CH2ONa+CH3CH3OH (b) CH3CCCH2CH2OH+Na+NH2NH3(l)arrow_forwardHistamine, whose release in the body triggers nasal secretions and constricted airways, has three nitrogen atoms. List them in order of increasing basicity and explain your ordering.arrow_forwardAnswer true or false to the following statements about the mechanism of acid-base reactions. Q) During an acid-base reaction the lone pair on the base fills the A-H antibonding sigma orbital.arrow_forward
- Which of the following has the strongest tendency to receive a H+? CH3COOH, Ka = 1.8 × 10−5 HCOOH, Ka = 1.8 × 10−4 Group of answer choices HCOO− CH3COOH CH3COO− HCOOHarrow_forwardArrange the compounds in each set in order of increasing acid strength. consult Table 4.1 for pKa values of each acid.arrow_forwardWhich of the following bases would abstract the hydroxyl hydrogen from (CH3)2CHOH? a. KH b. NaHCO3 c. NaOH d. Na2CO3 e. H2Oarrow_forward
- 1. Rank the following species in order of increasing acidity. Explain your reasons for ordering them as you do. HF NH3 H2SO4 CH3OH CH3COOH H3O+ H2O2. Consider the following compounds that vary from nearly nonacidic to strongly acidic. Draw the conjugate bases of these compounds, and explain why the acidity increases so dramatically with substitution by nitro groups. CH4 CH3NO2 CH2(NO2)2 CH(NO2)3arrow_forwardcalculate the lambda max Base Table: Heteroannular - 214nm Homoannular - 253nm Butadiene System - 217nm Acyclic trienes - 245nm alkyl +5 secondary amine +60 extending conjuction +30 exocyclic double bond +5 acetate +0 ether +6 thioether +30 double bond +5 Br, Cl +5arrow_forward3. Circle the most basic and put an X over the least basic in each set of three aminesarrow_forward

 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning



