
Chemistry: An Atoms First Approach
2nd Edition
ISBN: 9781305079243
Author: Steven S. Zumdahl, Susan A. Zumdahl
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 14, Problem 115CP
The titration of Na2CO3 with HCl bas the following qualitative profile:
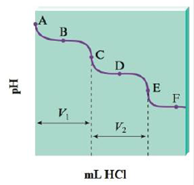
a. Identify the major species in solution at points A–F.
b. Calculate the pH at the halfway points to equivalence, B and D. (Hint: Refer to Exercise 113.)
Expert Solution & Answer
Trending nowThis is a popular solution!

Chapter 14 Solutions
Chemistry: An Atoms First Approach
Ch. 14 - What is meant by the presence of a common ion? How...Ch. 14 - Define a buffer solution. What makes up a buffer...Ch. 14 - Prob. 3RQCh. 14 - A good buffer generally contains relatively equal...Ch. 14 - Prob. 5RQCh. 14 - Prob. 6RQCh. 14 - Sketch the titration curve for a weak acid...Ch. 14 - Sketch the titration curve for a weak base...Ch. 14 - What is an acidbase indicator? Define the...Ch. 14 - Prob. 10RQ
Ch. 14 - What are the major species in solution after...Ch. 14 - Prob. 2ALQCh. 14 - Prob. 3ALQCh. 14 - Prob. 4ALQCh. 14 - Sketch two pH curves, one for the titration of a...Ch. 14 - Prob. 6ALQCh. 14 - Prob. 7ALQCh. 14 - You have a solution of the weak acid HA and add...Ch. 14 - The common ion effect for weak acids is to...Ch. 14 - Prob. 10QCh. 14 - Prob. 11QCh. 14 - Consider the following pH curves for 100.0 mL of...Ch. 14 - An acid is titrated with NaOH. The following...Ch. 14 - Consider the following four titrations. i. 100.0...Ch. 14 - Prob. 15QCh. 14 - Prob. 16QCh. 14 - How many of the following are buffered solutions?...Ch. 14 - Which of the following can be classified as buffer...Ch. 14 - A certain buffer is made by dissolving NaHCO3 and...Ch. 14 - Prob. 20ECh. 14 - Calculate the pH of each of the following...Ch. 14 - Calculate the pH of each of the following...Ch. 14 - Prob. 23ECh. 14 - Compare the percent ionization of the base in...Ch. 14 - Prob. 25ECh. 14 - Calculate the pH after 0.020 mole of HCl is added...Ch. 14 - Calculate the pH after 0.020 mole of NaOH is added...Ch. 14 - Calculate the pH after 0.020 mole of NaOH is added...Ch. 14 - Which of the solutions in Exercise 21 shows the...Ch. 14 - Prob. 30ECh. 14 - Calculate the pH of a solution that is 1.00 M HNO2...Ch. 14 - Calculate the pH of a solution that is 0.60 M HF...Ch. 14 - Calculate the pH after 0.10 mole of NaOH is added...Ch. 14 - Calculate the pH after 0.10 mole of NaOH is added...Ch. 14 - Calculate the pH of each of the following buffered...Ch. 14 - Prob. 36ECh. 14 - Calculate the pH of a buffered solution prepared...Ch. 14 - A buffered solution is made by adding 50.0 g NH4Cl...Ch. 14 - Prob. 39ECh. 14 - An aqueous solution contains dissolved C6H5NH3Cl...Ch. 14 - Prob. 41ECh. 14 - Prob. 42ECh. 14 - Consider a solution that contains both C5H5N and...Ch. 14 - Calculate the ratio [NH3]/[NH4+] in...Ch. 14 - Prob. 45ECh. 14 - Prob. 46ECh. 14 - Prob. 47ECh. 14 - Prob. 48ECh. 14 - Calculate the pH of a solution that is 0.40 M...Ch. 14 - Calculate the pH of a solution that is 0.20 M HOCl...Ch. 14 - Which of the following mixtures would result in...Ch. 14 - Prob. 52ECh. 14 - Prob. 53ECh. 14 - Calculate the number of moles of HCl(g) that must...Ch. 14 - Consider the titration of a generic weak acid HA...Ch. 14 - Sketch the titration curve for the titration of a...Ch. 14 - Consider the titration of 40.0 mL of 0.200 M HClO4...Ch. 14 - Consider the titration of 80.0 mL of 0.100 M...Ch. 14 - Consider the titration of 100.0 mL of 0.200 M...Ch. 14 - Prob. 60ECh. 14 - Lactic acid is a common by-product of cellular...Ch. 14 - Repeat the procedure in Exercise 61, but for the...Ch. 14 - Repeat the procedure in Exercise 61, but for the...Ch. 14 - Repeat the procedure in Exercise 61, but for the...Ch. 14 - Prob. 65ECh. 14 - In the titration of 50.0 mL of 1.0 M methylamine,...Ch. 14 - You have 75.0 mL of 0.10 M HA. After adding 30.0...Ch. 14 - A student dissolves 0.0100 mole of an unknown weak...Ch. 14 - Prob. 69ECh. 14 - Prob. 70ECh. 14 - Potassium hydrogen phthalate, known as KHP (molar...Ch. 14 - A certain indicator HIn has a pKa of 3.00 and a...Ch. 14 - Prob. 73ECh. 14 - Prob. 74ECh. 14 - Prob. 75ECh. 14 - Prob. 76ECh. 14 - Prob. 77ECh. 14 - Estimate the pH of a solution in which crystal...Ch. 14 - Prob. 79ECh. 14 - Prob. 80ECh. 14 - Prob. 81AECh. 14 - Prob. 82AECh. 14 - Tris(hydroxymethyl)aminomethane, commonly called...Ch. 14 - Prob. 84AECh. 14 - You have the following reagents on hand: Solids...Ch. 14 - Prob. 86AECh. 14 - Prob. 87AECh. 14 - What quantity (moles) of HCl(g) must be added to...Ch. 14 - Calculate the value of the equilibrium constant...Ch. 14 - The following plot shows the pH curves for the...Ch. 14 - Calculate the volume of 1.50 102 M NaOH that must...Ch. 14 - Prob. 92AECh. 14 - A certain acetic acid solution has pH = 2.68....Ch. 14 - A 0.210-g sample of an acid (molar mass = 192...Ch. 14 - The active ingredient in aspirin is...Ch. 14 - One method for determining the purity of aspirin...Ch. 14 - A student intends to titrate a solution of a weak...Ch. 14 - Prob. 98AECh. 14 - Prob. 99AECh. 14 - Consider 1.0 L of a solution that is 0.85 M HOC6H5...Ch. 14 - Prob. 101CWPCh. 14 - Consider the following acids and bases: HCO2H Ka =...Ch. 14 - Prob. 103CWPCh. 14 - Prob. 104CWPCh. 14 - Consider the titration of 100.0 mL of 0.100 M HCN...Ch. 14 - Consider the titration of 100.0 mL of 0.200 M...Ch. 14 - Prob. 107CWPCh. 14 - Prob. 108CPCh. 14 - A buffer is made using 45.0 mL of 0.750 M HC3H5O2...Ch. 14 - A 0.400-M solution of ammonia was titrated with...Ch. 14 - Prob. 111CPCh. 14 - Consider a solution formed by mixing 50.0 mL of...Ch. 14 - When a diprotic acid, H2A, is titrated with NaOH,...Ch. 14 - Consider the following two acids: In two separate...Ch. 14 - The titration of Na2CO3 with HCl bas the following...Ch. 14 - Prob. 116CPCh. 14 - A few drops of each of the indicators shown in the...Ch. 14 - Malonic acid (HO2CCH2CO2H) is a diprotic acid. In...Ch. 14 - A buffer solution is prepared by mixing 75.0 mL of...Ch. 14 - A 10.00-g sample of the ionic compound NaA, where...Ch. 14 - Prob. 121IPCh. 14 - Prob. 122MP
Additional Science Textbook Solutions
Find more solutions based on key concepts
The method to determine the volume of a powered solid, liquid and a rock needs to be determined. Concept introd...
Living By Chemistry: First Edition Textbook
Determine the number of protons, neutrons, and electrons in the following atoms: a. a hydrogen atom that has a ...
General, Organic, and Biological Chemistry (3rd Edition)
The chapter sections to review are shown in parentheses at the end of each problem. A "chemical-free” shampoo i...
Basic Chemistry
Q2. Which statement best defines chemistry?
a. The science that studies solvents, drugs, and insecticides
b. Th...
Introductory Chemistry (6th Edition)
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Without doing detailed calculations, sketch the curve for the titration of 30.0 mL of 0.10 M NaOH with 0.10 M HCl. Indicate the approximate pH at the beginning of the titration and at the equivalence point. What is the total solution volume at the equivalence point?arrow_forwardConsider a 10.0% (by mass) solution of hypochlorous acid. Assume the density of the solution to be 1.00 g/mL. A 30.0-mL sample of the solution is titrated with 0.419 M KOH. Calculate the pH of the solution (a) before titration. (b) halfway to the equivalence point. (c) at the equivalence point.arrow_forwardMalonic acid (HO2CCH2CO2H) is a diprotic acid. In the titration of malonic acid w ith NaOH, stoichiometric points occur at pH = 3.9 and 8.8. A 25.00-mL sample of malonic acid of unknown concentration is titrated with 0.0984 M NaOH, requiring 31.50 mL of the NaOH solution to reach the phenolphthalein end point. Calculate the concentration of the initial malonic acid solution. (Sec Exercise 113.)arrow_forward
- In an acid-base titration, 21.16 mL of an NaOH solution are needed to neutralize 20.04 mL of a 0.0997 M HCl solution. To find the molarity of the NaOH solution, we can use the following procedure: First note the value of MH in the HCl solution. ____________M Find MOH- in the NaOH solution. Use Eq.3. ____________M Obtain MNaOH from MOH. ____________Marrow_forwardThe titration of Na2CO3 with HCl bas the following qualitative profile: a. Identify the major species in solution at points AF. b. Calculate the pH at the halfway points to equivalence, B and D. (Hint: Refer to Exercise 113.)arrow_forwardA 0.400-M solution of ammonia was titrated with hydrochloric acid to the equivalence point, where the total volume was 1.50 times the original volume. At what pH does the equivalence point occur?arrow_forward
- Repeat the procedure in Exercise 61, but for the titration of 25.0 mL of 0.100 M NH3 (Kb = 1.8 105) with 0.100 M HCl.arrow_forwardA diprotic acid, H2B(MM=126g/moL), is determined to be a hydrate, H2B xH2O. A 10.00-g sample of this hydrate is dissolved in enough water to make 150.0 mL of solution. Twenty-five milliliters of this solution requires 48.5 mL of 0.425 M NaOH to reach the equivalence point. What is x?arrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you

 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Chemical Principles in the LaboratoryChemistryISBN:9781305264434Author:Emil Slowinski, Wayne C. Wolsey, Robert RossiPublisher:Brooks Cole
Chemical Principles in the LaboratoryChemistryISBN:9781305264434Author:Emil Slowinski, Wayne C. Wolsey, Robert RossiPublisher:Brooks Cole


Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:9781305079243
Author:Steven S. Zumdahl, Susan A. Zumdahl
Publisher:Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Chemical Principles in the Laboratory
Chemistry
ISBN:9781305264434
Author:Emil Slowinski, Wayne C. Wolsey, Robert Rossi
Publisher:Brooks Cole

Solutions: Crash Course Chemistry #27; Author: Crash Course;https://www.youtube.com/watch?v=9h2f1Bjr0p4;License: Standard YouTube License, CC-BY