
Concept explainers
(a)
Interpretation:
The IUPAC name for the given structure has to be written.
Concept introduction:
Any organic molecule can be named by using certain rules given by IUPAC (International Union for Pure and applied chemistry). IUPAC name consists of three parts, namely Prefix, suffix and root word.
Prefix- Represents the substituent present in the molecule and its position in the root name.
Suffix- Denotes the presence of
Root word - Represents the longest continuous carbon skeleton of the organic molecule.
(a)
Answer to Problem 14.21UKC
The name of the compound is 5-Methyl-3-hexanol.
Explanation of Solution
The name of the alcohol is given below,
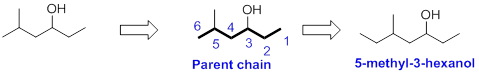
The number of carbon in parent chain is six, hence it is called as hexane. The functional group of the given molecule is alcohol therefore the suffix “ane” is replaced with “ol” indicating the presence of alcohol. In the given structure the third carbon is bonded with alcohol
(b)
Interpretation:
The IUPAC name for the given structure has to be written.
Concept introduction:
Naming
- (1) Substituted benzenes are named using ‘-benzene’ as the parent.
- (2) When benzene has more than one substituent, the position of those substituents is indicated by numbers or by ortho(o-), meta(m-) or para(p-).
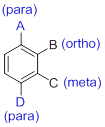
(b)
Answer to Problem 14.21UKC
The name of the compound is 1-Methoxy-3-methylbenzene.
Explanation of Solution
The name of the compound is given below,
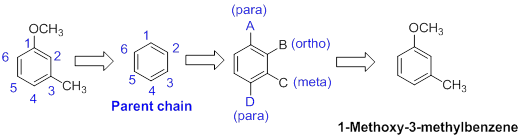
The parent ring is benzene. The functional group of the given molecule is methoxy and methyl group. The location of methoxy is in the first carbon and methyl group is third carbon of the benzene ring. Therefore the name of the molecule is 1-Methoxy-3-methylbenzene.
(c)
Interpretation:
The IUPAC name for the given structure has to be written.
Concept introduction:
Any organic molecule can be named by using certain rules given by IUPAC (International Union for Pure and applied chemistry). IUPAC name consists of three parts, namely Prefix, suffix and root word.
Prefix- Represents the substituent present in the molecule and its position in the root name.
Suffix- Denotes the presence of functional group if any in the molecule. It can be an alkene, alkyne, alcohol, carboxylic acid, alcohol etc. To add suffix to name a compound, the suffix “-ane” in the parent alkane is replaced by the respective suffix, which corresponds to the functional group present in the given compound. For carboxylic acid, suffix “-oic” will be added, for alcohol, suffix “-ol” will be added and so on
Root word - Represents the longest continuous carbon skeleton of the organic molecule.
(c)
Answer to Problem 14.21UKC
The name of the compound is 3-Methylcyclohexanol.
Explanation of Solution
The name of the compound is given below,
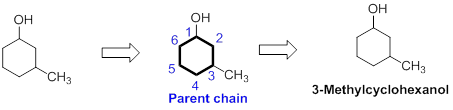
The number of carbon in parent chain is six and it is in the form of cycle, hence it is called as cyclohexane. The functional group of the given molecule is alcohol therefore the suffix “ane” is replaced with “ol” indicating the presence of alcohol. In the given structure, the first carbon is bonded with alcohol and third carbon is bonded with methyl
Want to see more full solutions like this?
Chapter 14 Solutions
Fundamentals of General, Organic, and Biological Chemistry, Books a la Carte Edition; Modified Mastering Chemistry with Pearson eText -- ValuePack ... and Biological Chemistry (4th Edition)
- Draw the structures of the following compounds. (Includes both new and old names.) 3-cyclopentylhexan-3-olarrow_forwardConsider the following acids and their ionization constant, determine which conjugate base is HCOOH Ka = 1.7 x 10-4 (b) HCN Ka = 4.9 x 10-10arrow_forwardIdentify the names of the following structure. (with alpha/beta and L-D designation)arrow_forward
- If the phosphorus atom in 3-phosphoglycerate is radioactively labeled, where will the label be when the reaction that forms 2-phosphoglycerate is over?arrow_forwardDraw condensed structural formulas for the two carboxylic acids with the molecular formula C4H8O2arrow_forwardWhat is the empirical formula for C3H6O3? C3H6O3 C6H12O6 CH2O None of thesearrow_forward
 BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman
BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman
Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY
Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON
Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON





