
Chemistry & Chemical Reactivity
9th Edition
ISBN: 9781133949640
Author: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 14, Problem 6PS
Phenyl acetate, an ester, reacts with water according to the equation
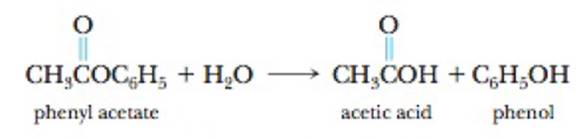
The data in the table were collected for this reaction at 5°C.
| Time (s) | [Phenyl acetate] (mol/L) |
| 0 | 0.55 |
| 15.0 | 0.42 |
| 30.0 | 0.31 |
| 45.0 | 0.23 |
| 60.0 | 0.17 |
| 75.0 | 0.12 |
| 90.0 | 0.085 |
- (a) Plot the phenyl acetate concentration versus time, and describe the shape of the curve observed.
- (b) Calculate the rate of change of the phenyl acetate concentration during the period 15.0 seconds to 30.0 seconds and also during the period 75.0 seconds to 90.0 seconds. Why is one value smaller than the other?
- (c) What is the rate of change of the phenyl acetate concentration during the time period 60.0 seconds to 75.0 seconds?
- (d) What is the instantaneous rate at 15.0 seconds?
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
You are synthesizing one fruity ester (pick any - does not have to be on the picture) for a special coffee. The challenge is that all alcohols and carboxylic acids are out of stock. Fortunately, any other chemical (e.g. methane) can be ordered. You will always find a suitable catalyst for any reaction.
Draw a flowchart to summarize the chemical reactions that will lead to the synthesis of this fruity ester. The fewer the steps the better. Describe each reaction in terms of necessary reactants and the type of reaction (see example below). Supply the IUPAC names of all organic compounds. No wall of text please. Diagrams are very welcome.
The breakdown of a certain pollutant X in sunlight is known to follow first-order kinetics. An atmospheric scientist studying the process fills a 15.0 L reaction flask with a sample of urban air and finds that the partlal pressure of X In the flask decreases from 0.115 atm to 0.0753 atm over 8.7 hours. Calculate the initial rate of decomposition of X, that is, the rate at which X was disappearing at the start of the experiment. Round your answer to 2 significant digits.
The Nobel Prize in Chemistry was awarded in 1995 to Paul Crutzen, Mario Molina and Sherwood Rowland for research showing the chlorofluorcarbons (CFCs) were catalyzing the destruction of ozone in the stratosphere. The catalytic process can be described by a two step mechanism:
(1) Cl + O3 --> ClO + O2 (slow)
(2) ClO + O --> Cl + O2 (fast)
Determine the overall reaction for this mechanism, and answer the following question:
In this mechanism Cl acts as a(n) ______________ and ClO acts as a(n) ___________________.
a.
reagent, intermediate
b.
intermediate, catalyst
c.
catalyst, intermediate
d.
intermediate, product
Chapter 14 Solutions
Chemistry & Chemical Reactivity
Ch. 14.1 - Sucrose decomposes to fructose and glucose in acid...Ch. 14.1 - What are the relative rates of appearance or...Ch. 14.1 - Prob. 1RCCh. 14.1 - 2. Use the graph provided in Example 14.1 to...Ch. 14.2 - 1. Which of the following will not usually...Ch. 14.3 - The initial rate ( [NO]/ t] of the reaction of...Ch. 14.3 - The rate constant, k, at 25 C is 0.27/h for the...Ch. 14.3 - The reaction NO(g) + 1/2 Cl2(g) NOCl(g) is...Ch. 14.4 - Sucrose, a sugar, decomposes in acid solution to...Ch. 14.4 - Gaseous azomethane (CH3N2CH3) decomposes to ethane...
Ch. 14.4 - Prob. 3CYUCh. 14.4 - The catalyzed decomposition of hydrogen peroxide...Ch. 14.4 - Americium is used in smoke detectors and in...Ch. 14.4 - The decomposition of N2O5 is a first-order...Ch. 14.4 - Which of the following will confirm that the...Ch. 14.4 - 3. The equation for the decomposition of NO2(g) at...Ch. 14.5 - Prob. 1CYUCh. 14.5 - The colorless gas N2O4, decomposes to the brown...Ch. 14.5 - Prob. 1RCCh. 14.5 - Prob. 2RCCh. 14.6 - Nitrogen monoxide is reduced by hydrogen to give...Ch. 14.6 - Prob. 2CYUCh. 14.6 - One possible mechanism for the decomposition of...Ch. 14.6 - The rate equation for a reaction A + B C was...Ch. 14.6 - A reaction is believed to occur by the following...Ch. 14.6 - Prob. 1QCh. 14.6 - Prob. 2QCh. 14.6 - Prob. 3QCh. 14.6 - Prob. 4QCh. 14.6 - Prob. 5QCh. 14.6 - Determine the activation energy for the reaction...Ch. 14 - Give the relative rates of disappearance of...Ch. 14 - Give the relative rates of disappearance of...Ch. 14 - In the reaction 2 O3(g) 3 O2(g), the rate of...Ch. 14 - In the synthesis of ammonia, if [H2]/t = 4.5 104...Ch. 14 - Experimental data are listed here for the reaction...Ch. 14 - 6. Phenyl acetate, an ester, reacts with water...Ch. 14 - Using the rate equation Rate = k[A]2[B], define...Ch. 14 - A reaction has the experimental rate equation Rate...Ch. 14 - The reaction between ozone and nitrogen dioxide at...Ch. 14 - Nitrosyl bromide, NOBr, is formed from NO and Br2:...Ch. 14 - The data in the table are for the reaction of NO...Ch. 14 - The reaction 2 NO(g) + 2 H2(g) N2(g) + 2 H2O(g)...Ch. 14 - Data for the reaction NO(g) + O2(g) NO2(g) are...Ch. 14 - Data for the following reaction are given in the...Ch. 14 - The rate equation for the hydrolysis of sucrose to...Ch. 14 - The decomposition of N2O5 in CCl4 is a first-order...Ch. 14 - The decomposition of SO2Cl2 is a first-order...Ch. 14 - The conversion of cyclopropane to propene (Example...Ch. 14 - Hydrogen peroxide, H2O2(aq), decomposes to H2O()...Ch. 14 - The decomposition of nitrogen dioxide at a high...Ch. 14 - At 573 K, gaseous NO2(g) decomposes, forming NO(g)...Ch. 14 - The dimerization of butadiene, C4H6, to form...Ch. 14 - The decomposition of ammonia on a metal surface to...Ch. 14 - Hydrogen iodide decomposes when heated, forming...Ch. 14 - The rate equation for the decomposition of N2O5...Ch. 14 - Gaseous azomethane, CH3N=NCH3, decomposes in a...Ch. 14 - The decomposition of SO2Cl2 SO2Cl2(g) SO2(g) +...Ch. 14 - The compound Xe(CF3)2 decomposes in a first-order...Ch. 14 - The radioactive isotope 64Cu is used in the form...Ch. 14 - Radioactive gold-198 is used in the diagnosis of...Ch. 14 - Prob. 31PSCh. 14 - Ammonia decomposes when heated according to the...Ch. 14 - Gaseous NO2 decomposes at 573 K. NO2(g) NO(g) + ...Ch. 14 - The decomposition of HOF occurs at 25 C. HOF(g) ...Ch. 14 - Prob. 35PSCh. 14 - Prob. 36PSCh. 14 - Calculate the activation energy, Ea, for the...Ch. 14 - If the rate constant for a reaction triples when...Ch. 14 - When healed lo a high temperature, cyclobutane,...Ch. 14 - When heated, cyclopropane is converted to propene...Ch. 14 - The reaction of H2 molecules with F atoms H2(g) +...Ch. 14 - Prob. 42PSCh. 14 - What is the rate law for each of the following...Ch. 14 - What is the rate law for each of the following...Ch. 14 - Ozone, O3, in the Earths upper atmosphere...Ch. 14 - The reaction of NO2(g) and CO(g) is thought to...Ch. 14 - A proposed mechanism for the reaction of NO2 and...Ch. 14 - The mechanism for the reaction of CH3OH and HBr is...Ch. 14 - A reaction has the following experimental rate...Ch. 14 - For a first-order reaction, what fraction of...Ch. 14 - Prob. 51GQCh. 14 - Data for the following reaction are given in the...Ch. 14 - Formic acid decomposes at 550 C according to the...Ch. 14 - Isomerization of CH3NC occurs slowly when CH3NC is...Ch. 14 - When heated, tetrafluoroethylene dimerizes to form...Ch. 14 - Data in the table were collected at 540 K for the...Ch. 14 - Ammonium cyanate, NH4NCO, rearranges in water to...Ch. 14 - Prob. 58GQCh. 14 - At temperatures below 500 K, the reaction between...Ch. 14 - Nitryl fluoride can be made by treating nitrogen...Ch. 14 - The decomposition of dinitrogen pentaoxide N2O5(g)...Ch. 14 - The data in the table give the temperature...Ch. 14 - The decomposition of gaseous dimethyl ether at...Ch. 14 - The decomposition of phosphine, PH3, proceeds...Ch. 14 - The thermal decomposition of diacetylene, C4H2,...Ch. 14 - Prob. 66GQCh. 14 - The ozone in the Earths ozone layer decomposes...Ch. 14 - Hundreds of different reactions occur in the...Ch. 14 - Data for the reaction [Mn(CO)5(CH3CN)]+ + NC5H5 ...Ch. 14 - The gas-phase reaction 2 N2O5(g) 4 NO2(g) + O2(g)...Ch. 14 - Prob. 71GQCh. 14 - The decomposition of SO2Cl2 to SO2 and Cl2 is...Ch. 14 - The decomposition of nitrogen dioxide at a high...Ch. 14 - Prob. 74GQCh. 14 - Egg protein albumin is precipitated when an egg is...Ch. 14 - A The compound 1,3-butadiene (C4H6) forms...Ch. 14 - Hypofluorous acid, HOF, is very unstable,...Ch. 14 - We know that the decomposition of SO2Cl2 is...Ch. 14 - Nitramide, NO2NH2, decomposes slowly in aqueous...Ch. 14 - Prob. 80GQCh. 14 - Prob. 83ILCh. 14 - Prob. 84ILCh. 14 - The oxidation of iodide ion by the hypochlorite...Ch. 14 - The acid-catalyzed iodination of acetone...Ch. 14 - Prob. 87SCQCh. 14 - The following statements relate to the reaction...Ch. 14 - Chlorine atoms contribute to the destruction of...Ch. 14 - Prob. 91SCQCh. 14 - Prob. 92SCQCh. 14 - The reaction cyclopropane propene occurs on a...Ch. 14 - Prob. 94SCQCh. 14 - Examine the reaction coordinate diagram given...Ch. 14 - Draw a reaction coordinate diagram for an...
Additional Science Textbook Solutions
Find more solutions based on key concepts
16.43 The following pictures represent solutions at various stages in thetitration of a weak diprotic acid with...
Chemistry (7th Edition)
Q2. Which statement best defines chemistry?
a. The science that studies solvents, drugs, and insecticides
b. Th...
Introductory Chemistry (5th Edition) (Standalone Book)
How could you separate a mixture of the following compounds? The reagents available to you are water, either, 1...
Organic Chemistry
For each of the following 2-dimensional shapes, determine the highest order rotation axis of symmetry.
Inorganic Chemistry
Label each statement about the polynucleotide ATGGCG as true or false. The polynucleotide has six nucleotides. ...
General, Organic, & Biological Chemistry
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Sketch the reaction mechanism (including the final product) corresponding to the following description. Be prepared to explain the mechanism to your partner. 2-bromo-4-methylpentane is being reacted with sodium cyanide in diethyl ether (set up the reaction – next you will get a description of the mechanism to draw). A lone pair of electrons from the cyanide attacks the carbon bonded to bromine, forming a new carbon-carbon bond. At the same time, the carbon-bromine bond breaks with the electron pair from the carbon-bromine bond moving toward the bromine to form a bromide ion. This will form the product 2,4-dimethylpentanenitrile and sodium bromide. is this reaction following an SN1 or SN2 mechanism? Write out the rate law for this reaction.Draw a reaction coordinate diagram for this reaction.arrow_forwardhow reaction rate increases in pressure cooking because of collision theory and energy potential diagrams : how the rate of reaction is affected by temperature, surface area, nature of reactants, concentration, etc. by using collision theory? (use Hess’s Law, Le Châtelier’s Principle, or Kinetic Molecular Theory to explain in increasing rate of reaction and relevance by including all 4 required subtopics )arrow_forwardRuns [S2O8 2-], M [I–], M [S2O3 2-], M Reaction time,s Rate, M/s 1 49.10 2 106.10 3 197.44 4 69.20 5 53.05 please complete the tablearrow_forward
- Which statement best describes the image? This image illustrates the calculation of an instantaneous rate for the product. This image illustrates the calculation of an average rate for the product. This image illustrates the calculation of an average rate for the product. This image illustrates the calculation of an instantaneous rate for the reactant.arrow_forwardBased on the theory of particles and collisions between molecules, explain the effect that there will be a decrease in temperature, a decrease in the pressure of the reagents and a increasing the contact area over the rate of a chemical reaction.arrow_forwardIn 30.0 minutes, a first order reaction is 25% complete. Calculate how long it will take for the reaction to be 95.0% complete.arrow_forward
- The following data are for the rearrangement of cyclopropane to propene at 500 °C.(CH2)3 CH3CH=CH2 [ (CH2)3 ], M 8.65×10-2 4.33×10-2 2.17×10-2 1.09×10-2 time, min 0 12.2 24.4 36.6 Hint: It is not necessary to graph these data.(1)The half life observed for this reaction is min .(2)Based on these data, the rate constant for this order reaction is min -1.arrow_forwardBased on the 1st order kinetic and Langmuir adsorption isotherm to derive the overall rate equation for the following catalytic reactions.assume your own hypothesis hypothesis Overall Reaction CO+(1⁄2)02 CO2 Step 1: CO+2CuO Cu2O + CO2 Step 2: Cu2O+FeO2CuO + Fe Step 3: (1/2)O2+ Fe FeOarrow_forwardSketch the reaction mechanism (including the final product) corresponding to the following description of; 2-bromo-4-methylpentane is being reacted with sodium cyanide in diethyl ether (set up the reaction – next you will get a description of the mechanism to draw). A lone pair of electrons from the cyanide attacks the carbon bonded to bromine, forming a new carbon-carbon bond. At the same time, the carbon-bromine bond breaks with the electron pair from the carbon-bromine bond moving toward the bromine to form a bromide ion. This will form the product 2,4-dimethylpentanenitrile and sodium bromide.arrow_forward
- Ozone can oxidize HSO3^1-(aq) as represented below: HSO3^-(aq)+03(aq)---> HSO4^-(aq) +02(g) A solution is prepared in which the initial concentration of HSO3^1- (aq)(6.4x10^-4M) is much larger than that of O3(aq)(1.0x10^-5M). The concentration of O3(aq) is monitored as the reaction proceeds, and the data are plotted in the graph below: (see attached image) iii)Considering the relative concentration of the reactants, briefly explain why the data in the graph are also consistent with the following rate law: Rate=k1[O3][HSO3^1-] iv)Briefly describe an experiment that could provide evidence to support the rate law given in part (diii)arrow_forwardState whether the statements are true or false. 1. The instantaneous reaction is always equal and constant. 2. Similar elements tend to react similarly, but may do so at different reaction rates 3. The rate law for a chemical reaction is always expressed as a product of the initial concentration of products of the reaction. 4. Chemical entities can be in any orientation during an effective solution. 5. The reaction mechanism explains exactly what occurs in a chemical reaction. 6. It is not necessary to break existing bonds in molecules in order to form new bonds. 7. The frequency of collisions and the proportion of those collisions that are effective determine the rate of the given reactionarrow_forwardWhen the concentration of reactant molecules is increased, the rate of reaction increases. The best explanation is: As the reactant concentration increases, the rate constant increases. the activation energy increases. the frequency of molecular collisions increases. the order of reaction increases. the average kinetic energy of molecules increases.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning
Kinetics: Chemistry's Demolition Derby - Crash Course Chemistry #32; Author: Crash Course;https://www.youtube.com/watch?v=7qOFtL3VEBc;License: Standard YouTube License, CC-BY