
Concept explainers
a.
Interpretation:
Compound that is formed when the given aldose is treated with hydrogen in presence of palladium as catalyst has to be drawn.
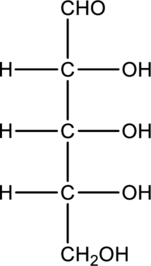
Concept Introduction:
Carbohydrates undergo reduction similar to an
b.
Interpretation:
Compound that is formed when the given aldose is treated with hydrogen in presence of palladium as catalyst has to be drawn.
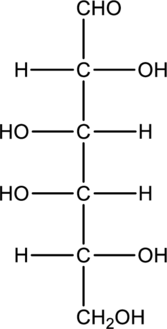
Concept Introduction:
Refer to part “a.”.
c.
Interpretation:
Compound that is formed when the given aldose is treated with hydrogen in presence of palladium as catalyst has to be drawn.
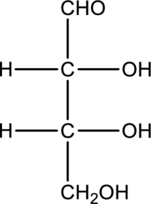
Concept Introduction:
Refer to part “a.”.
Want to see the full answer?
Check out a sample textbook solution
Chapter 14 Solutions
Principles of General, Organic, Biological Chemistry
- Which D-aldopentose is oxidized to an optically active aldaric acid and undergoes the Wohl degradation to yield a D-aldotetrose that is oxidized to an optically active aldaric acid?arrow_forwardWhich aldoses are oxidized to optically inactive aldaric acids: (a) D-erythrose; (b) Dlyxose; (c) D-galactose?arrow_forwardDraw the product formed when CH3CH2C=CH is treated with each of the following sets of reagents: (a) H2O, H2SO4, HgSO4; and (b) R2BH, followed by H2O2, HO−.arrow_forward
- What products are formed when each compound undergoes a Kiliani– Fischer synthesis?arrow_forwardConvert each aldohexose to the indicated anomer using a Haworth projection.arrow_forwardIdentify the reducing and non reducing sugars in the following. Which compounds, is any, will react with Tollen’s reagent?arrow_forward
- Draw the product formed when CH3CH2CH2CH2CH=CH2 is treated with either(a) H2O, H2SO4; or (b) BH3 followed by H2O2, HO−.arrow_forwardThe following isomerization reaction, drawn using D-glucose as starting material, occurs with all aldohexoses in the presence of base. Draw a stepwise mechanism that illustrates how each compound is formed.arrow_forwardDraw the products formed when β-D-galactose is treated with each reagent. a. Ag2O + CH3I b. NaH + C6H5CH2Cl c. The product in (b), then H3O+ d. Ac2O + pyridine e. C6H5COCl + pyridine f. The product in (c), then C6H5COCl + pyridinearrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





