
Interpretation:
The given cycloalkane undergoes reaction with the given Simmons-Smith reagent which is stereospecific and gives only the isomer shown. The reason has to be suggested.
Concept Introduction:
Simmons-Smith reaction: This is ultrasonication improve the rate of formation of these organic zinc compounds, as with many organometallic reactions occurring at a surface condition.
Example: The substance of a carbenoid a carbine like substance that converters
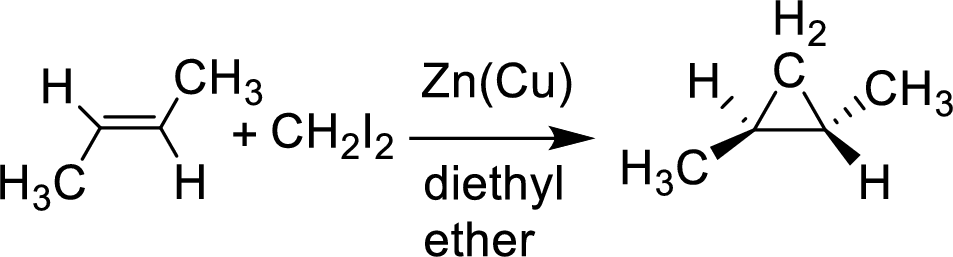
Addition Reaction:
- It is a type of reaction in which two reactants adding together to form a single product.
- It can be said as a reverse reaction of elimination reaction.
- It is a characteristic reaction of
alkane .
Hydrogenation reaction is an addition reaction in which addition of hydrogen to an unsaturated molecule occurs
Stereoisomers are isomeric molecules that have the same molecular formula and sequence of bonded atoms but differ in the three-dimensional orientations of their atoms in space.
Stereospecific reaction: reaction undergoes from a stereoisomer to a unique stereo isomeric product.
Trending nowThis is a popular solution!

Chapter 15 Solutions
Organic Chemistry
- we know that ethers, such as diethyl ether and tetrahydrofuran, are quite resistant to the action of dilute acids and require hot concentrated HI or HBr for cleavage. However, acetals in which two ether groups are linked to the same carbon undergo hydrolysis readily, even in dilute aqueous acid. How do you account for this marked difference in chemical reactivity toward dilute aqueous acid between ethers and acetals?arrow_forwardThe Williamson ether synthesis involves treatment of a haloalkane with a metal alkoxide. Following are two reactions intended to give benzyl tert-butyl ether. One reaction gives the ether in good yield, and the other reaction does not. Which reaction gives the ether? What is the product of the other reaction, and how do you account for its formation?arrow_forwardreaction of either 3-bromo-1-butene or (Z) -1-bromo-2-butene with water under SN1 condition yields the same product explain whyarrow_forward
- 1. Compound A, C9H12, absorbs 3 equivalents of H2 on catalytic hydrogenation over palladium catalyst to give B, C9H18. On the treatment with acidic KMnO4, compound A gives among other things, a ketone that was identified as cyclohexanone. On reaction with NaNH2 in NH3, followed by addition of iodomethane, compound A gives a new hydrocarbon C, C10H14. What are the structures of A , B and C?arrow_forwardWhen sodium methoxide base and hydrogen bromide are removed from trans-1-bromo 2-methyl cyclohexane, considering the conformation of the starting compound, find the product that will be formed by writing and explaining the mechanism of the reaction.arrow_forwardCyclobutane fracts with bromine to give bromocyclobutane, but bicyclobutane reacts with bromine to give 1,3-dibromocyclobutane. Account for the differences between the reactions of thee two compounds.arrow_forward
- In the dehydrohalogenation of bromocyclodecane, the major product is cis -cyclodecene rather than trans-cyclodecene. Offer an explanation.arrow_forwardHow can you distinguish between 1,3-cyclohexadiene and 1,4-cyclohexadiene by oxidative cleavage reactionsarrow_forwardwhy do (R)-3-chloro-1-methylcycloprop-1-ene and 5-Bromo-1,3-Cyclohexadiene react differently with cyanide (why doesn't cyanide pass through its usual mechanism for these two products and what did you obtain these specific products)arrow_forward
- What is the major product obtained from treating an excess of each of the following compounds with Cl2 in the presence of ultraviolet light at roomtemperature? Disregard stereoisomers.arrow_forwardShow stereo chemistry and possible outcomes and the steps for the reaction.arrow_forwardGive the product(s) of the reaction between 1-hexyne and each of the following reagents.Indicate regiochemistry and geometry (stereochemistry) where appropriate.a) 1 mole H2 and Lindlar’s catalyst: 1-hexeneb) 2 moles H2 and Pt: 1-hexanec) 2 moles Br2:d) 1 mole Br2e) 2 moles HClf) NaNH2 in NH3(l)g) HgSO4, H2SO4, H2Oh) Aqueous NaOHi) (Sia)2BH,THF followed by H2O2, HO- j) 2 mole Li in NH3(l)arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

