
CHEMISTRY: ATOMS FIRST VOL 1 W/CONNECT
14th Edition
ISBN: 9781259327933
Author: Burdge
Publisher: MCG
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 15.2, Problem 2PPC
Consider the reaction
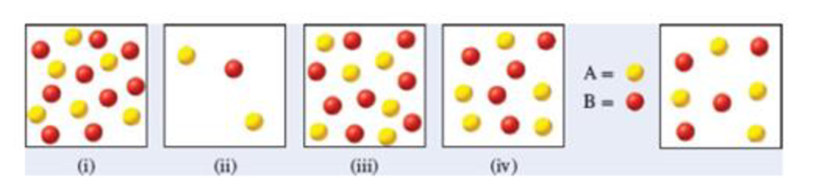
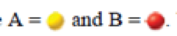 . Which of the following diagrams [(i)—(iv)] also represents a system at equilibrium? Select all that apply
. Which of the following diagrams [(i)—(iv)] also represents a system at equilibrium? Select all that apply
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Chapter 15 Solutions
CHEMISTRY: ATOMS FIRST VOL 1 W/CONNECT
Ch. 15.2 - Write reaction quotients for the following...Ch. 15.2 - Write the reaction quotient for each of the...Ch. 15.2 - Write the equation for the equilibrium that...Ch. 15.2 - In principle, in the reaction of A and B to form...Ch. 15.2 - Carbonyl chloride (COCl2), also called phosgene,...Ch. 15.2 - In an analysis of the following reaction at 100C....Ch. 15.2 - In another analysis at 100C involving the same...Ch. 15.2 - Consider the reaction 2AB. The diagram shown on...Ch. 15.2 - Prob. 15.2.1SRCh. 15.2 - Prob. 15.2.2SR
Ch. 15.3 - Write equilibrium expressions for each of the...Ch. 15.3 - Write equilibrium expressions for each of the...Ch. 15.3 - Which of the following equilibrium expressions...Ch. 15.3 - Consider the reaction A(s)+B(g)C(s). Which of the...Ch. 15.3 - The following reactions have the indicated...Ch. 15.3 - The following reactions have the indicated...Ch. 15.3 - Using the data from Practice Problem A, determine...Ch. 15.3 - Consider a chemical reaction represented by the...Ch. 15.3 - Write KP expressions for (a) PCl3(g) + Cl2(g) ...Ch. 15.3 - Write KP expressions for...Ch. 15.3 - Write the equation for the gaseous equilibrium...Ch. 15.3 - These diagrams represent closed systems at...Ch. 15.3 - The equilibrium constant, Kc, for the reaction...Ch. 15.3 - For the reaction N2(g)+3H2(g)2NH2(g) KC is 2.3 ...Ch. 15.3 - KP = 2.79 10-5 for the reaction in Practice...Ch. 15.3 - Consider the reaction 2A(l)2B(g) at room...Ch. 15.3 - Prob. 15.3.1SRCh. 15.3 - Prob. 15.3.2SRCh. 15.3 - Prob. 15.3.3SRCh. 15.3 - Prob. 15.3.4SRCh. 15.4 - At 375C, the equilibrium constant for the reaction...Ch. 15.4 - The equilibrium constant, Kc, for the formation of...Ch. 15.4 - Calculate KP for the formation of nitrosyl...Ch. 15.4 - Consider the reaction 2AB. The diagram shown on...Ch. 15.4 - The equilibrium constant, KP, for the reaction...Ch. 15.4 - G for the reaction H2(g)+I2(s)2HI(g) is 2.60...Ch. 15.4 - Prob. 8PPBCh. 15.4 - Prob. 8PPCCh. 15.4 - Using data from Appendix 2, calculate the...Ch. 15.4 - Prob. 9PPACh. 15.4 - Kf for the complex ion Ag(NH3)2+ is 1.5 107 at...Ch. 15.4 - Which of the following graphs [(i)(iv)] best shows...Ch. 15.4 - The equilibrium constant, Ksp, for the dissolution...Ch. 15.4 - Calculate G for the process:...Ch. 15.4 - Ksp for Co(OH)2 at 25C is 3.3 10-16 Using this and...Ch. 15.4 - Prob. 10PPCCh. 15.4 - Prob. 15.4.1SRCh. 15.4 - Prob. 15.4.2SRCh. 15.4 - Prob. 15.4.3SRCh. 15.5 - Kc for the reaction of hydrogen and iodine to...Ch. 15.5 - Calculate the equilibrium concentrations of H2,...Ch. 15.5 - Determine the initial concentration of HI if the...Ch. 15.5 - Consider the reaction A(g) + B(g) C(g). The...Ch. 15.5 - For the same reaction and temperature as in Worked...Ch. 15.5 - Prob. 12PPACh. 15.5 - Prob. 12PPBCh. 15.5 - Prob. 12PPCCh. 15.5 - At elevated temperatures, iodine molecules break...Ch. 15.5 - Aqueous hydrocyanic acid (HCN) ionizes according...Ch. 15.5 - Consider a weak acid, HA, that ionizes according...Ch. 15.5 - Prob. 13PPCCh. 15.5 - A mixture of 5.75 atm of H2 and 5.75 atm of I2 is...Ch. 15.5 - Prob. 14PPACh. 15.5 - Prob. 14PPBCh. 15.5 - Consider the reaction A(g)+B(g)C(s)+D(s). The...Ch. 15.5 - Prob. 15.5.1SRCh. 15.5 - Prob. 15.5.2SRCh. 15.5 - Prob. 15.5.3SRCh. 15.6 - Hydrogen sulfide (H2S) is a contaminant commonly...Ch. 15.6 - For each change indicated, determine whether the...Ch. 15.6 - What can be added to the equilibrium that will (a)...Ch. 15.6 - Consider the reaction A(g)+B(g)C(s)+D(s), of the...Ch. 15.6 - For each reaction, predict in what direction the...Ch. 15.6 - For each reaction, predict the direction of shift...Ch. 15.6 - For the following equilibrium, give an example of...Ch. 15.6 - Prob. 16PPCCh. 15.6 - Prob. 15.6.1SRCh. 15.6 - Prob. 15.6.2SRCh. 15.6 - Prob. 15.6.3SRCh. 15.6 - Prob. 15.6.4SRCh. 15 - Define equilibrium. Give two examples of a dynamic...Ch. 15 - Which of the following statements is collect about...Ch. 15 - Consider the reversible reaction A B. Explain how...Ch. 15 - What is the law of mass action?Ch. 15 - Briefly describe the importance of equilibrium in...Ch. 15 - Define reaction quotient. How does it differ from...Ch. 15 - Prob. 15.7QPCh. 15 - Write the equation for the reaction that...Ch. 15 - Prob. 15.9QPCh. 15 - Prob. 15.10QPCh. 15 - Prob. 15.11QPCh. 15 - The equilibrium constant for the reaction...Ch. 15 - Prob. 15.13QPCh. 15 - Prob. 15.14QPCh. 15 - Prob. 15.15QPCh. 15 - Prob. 15.16QPCh. 15 - Prob. 15.17QPCh. 15 - Write equilibrium constant expressions for Kc and...Ch. 15 - Write the equilibrium constant expressions for Kc...Ch. 15 - Prob. 15.20QPCh. 15 - Prob. 15.21QPCh. 15 - Prob. 15.22QPCh. 15 - Computational Problems 15.23 The equilibrium...Ch. 15 - Prob. 15.24QPCh. 15 - The equilibrium constant KP for the reaction is...Ch. 15 - Prob. 15.26QPCh. 15 - Prob. 15.27QPCh. 15 - Prob. 15.28QPCh. 15 - Prob. 15.29QPCh. 15 - The equilibrium constant Kp for foe reaction is...Ch. 15 - Ammonium carbamate (NH4CO2NH2) decomposes as...Ch. 15 - Prob. 15.32QPCh. 15 - Consider the equilibrium If nitrosyl bromide...Ch. 15 - Prob. 15.34QPCh. 15 - The following equilibrium constants have been...Ch. 15 - The following equilibrium constants were...Ch. 15 - At a certain temperature, the following reactions...Ch. 15 - Prob. 15.38QPCh. 15 - The equilibrium constant for the reaction A B is...Ch. 15 - Prob. 15.40QPCh. 15 - Explain why Equation 15.6 is of great importance...Ch. 15 - Fill in the missing entries in the following...Ch. 15 - Computational Problems 15.43 The aqueous reaction...Ch. 15 - For the autoionization of water at 25C,...Ch. 15 - Consider the following reaction at 25C....Ch. 15 - Prob. 15.46QPCh. 15 - (a) Calculate G and KP for the following...Ch. 15 - The equilibrium constant (KP) for the reaction...Ch. 15 - Consider the decomposition of calcium carbonate....Ch. 15 - The equilibrium constant KP for the reaction CO(g)...Ch. 15 - Prob. 15.51QPCh. 15 - Prob. 15.52QPCh. 15 - Prob. 15.53QPCh. 15 - Conceptual Problems 15.54 A and B react to form...Ch. 15 - If Kc. = 2 for the reaction A2 + B2 2AB at a...Ch. 15 - Prob. 15.1VCCh. 15 - Prob. 15.2VCCh. 15 - Prob. 15.3VCCh. 15 - Prob. 15.4VCCh. 15 - Review Questions Outline the steps for calculating...Ch. 15 - Prob. 15.57QPCh. 15 - Prob. 15.58QPCh. 15 - Prob. 15.59QPCh. 15 - The dissociation of molecular iodine into iodine...Ch. 15 - The equilibrium constant Kc for the decomposition...Ch. 15 - Consider the following equilibrium process at...Ch. 15 - Prob. 15.63QPCh. 15 - Prob. 15.64QPCh. 15 - Prob. 15.5VCCh. 15 - Prob. 15.6VCCh. 15 - Prob. 15.7VCCh. 15 - Prob. 15.8VCCh. 15 - Prob. 15.9VCCh. 15 - Prob. 15.10VCCh. 15 - Prob. 15.11VCCh. 15 - Prob. 15.12VCCh. 15 - Prob. 15.65QPCh. 15 - Prob. 15.66QPCh. 15 - Prob. 15.67QPCh. 15 - Conceptual Problems 15.68 Which of the following...Ch. 15 - For which of the following reactions will a change...Ch. 15 - Which of the following equilibria will shift to...Ch. 15 - Which of the following will cause the equilibrium...Ch. 15 - Consider the following equilibrium system...Ch. 15 - Heating solid sodium bicarbonate in a closed...Ch. 15 - Consider the following equilibrium systems....Ch. 15 - What effect does an increase in pressure have on...Ch. 15 - Prob. 15.76QPCh. 15 - Consider the following equilibrium process....Ch. 15 - Prob. 15.78QPCh. 15 - Consider the following equilibrium reaction in a...Ch. 15 - Consider the gas-phase reaction...Ch. 15 - Prob. 15.81QPCh. 15 - Prob. 15.82QPCh. 15 - Prob. 15.83QPCh. 15 - The simplified equation representing the binding...Ch. 15 - Prob. 15.85QPCh. 15 - ADDITIONAL PROBLEMS 15.86 Consider the following...Ch. 15 - The equilibrium constant Kp for the reaction...Ch. 15 - For a reaction with a negative G value, which of...Ch. 15 - Carbon monoxide (CO) and nitric oxide (NO) are...Ch. 15 - Consider the following reacting system....Ch. 15 - At a certain temperature and a total pressure of...Ch. 15 - The decomposition of ammonium hydrogen sulfide...Ch. 15 - Consider the reaction 2NO(g)+O2(g)2NO2(g) At 430C,...Ch. 15 - In the Mond process for the purification of...Ch. 15 - Consider the reaction N2(g)+O2(g)2NO(g) Given that...Ch. 15 - Prob. 15.96QPCh. 15 - A mixture of 0.47 mole of H2 and 3.59 moles of HCl...Ch. 15 - Prob. 15.98QPCh. 15 - The following reaction represents the removal of...Ch. 15 - Prob. 15.100QPCh. 15 - Prob. 15.101QPCh. 15 - Calculate the equilibrium pressure of CO2 due to...Ch. 15 - Prob. 15.103QPCh. 15 - Consider the gas-phase reaction between A2 (green)...Ch. 15 - Prob. 15.105QPCh. 15 - The following diagram represents a gas-phase...Ch. 15 - The formation of SO3 from SO2 and O2 is an...Ch. 15 - Calculate the pressure of O2 (in atm) over a...Ch. 15 - The following reaction was described as the cause...Ch. 15 - Prob. 15.110QPCh. 15 - Calculate G and Kp for the following processes at...Ch. 15 - Prob. 15.112QPCh. 15 - The equilibrium constant Kp for the following...Ch. 15 - Prob. 15.114QPCh. 15 - Prob. 15.115QPCh. 15 - Prob. 15.116QPCh. 15 - Prob. 15.117QPCh. 15 - Prob. 15.118QPCh. 15 - Prob. 15.119QPCh. 15 - Prob. 15.120QPCh. 15 - The equilibrium constant Kc for the reaction...Ch. 15 - For reactions earned out under standard-state...Ch. 15 - When a gas was heated under atmospheric...Ch. 15 - Prob. 15.124QPCh. 15 - The equilibrium constant Kc for the following...Ch. 15 - The equilibrium constant (KP for the formation of...Ch. 15 - Prob. 15.127QPCh. 15 - Prob. 15.128QPCh. 15 - Prob. 15.129QPCh. 15 - In the gas phase, nitrogen dioxide is actually a...Ch. 15 - A 2.50-mole sample of NOCl was initially in a...Ch. 15 - About 75% of hydrogen for industrial use is...Ch. 15 - Photosynthesis can be represented by...Ch. 15 - Consider the decomposition of ammonium chloride at...Ch. 15 - At 25C, the equilibrium partial pressures of NO2...Ch. 15 - In 1899 the German chemist Ludwig Mond developed a...Ch. 15 - Consider the equilibrium reaction described in...Ch. 15 - Consider the equilibrium system3AB. Sketch the...Ch. 15 - The vapor pressure of mercury is 0.0020 mmHg at...Ch. 15 - Large quantities of hydrogen are needed for the...Ch. 15 - Prob. 15.141QPCh. 15 - At 25C. a mixture of NO2 and N2O4 gases are m...Ch. 15 - Prob. 15.143QPCh. 15 - Heating copper (II) oxide at 400C does not produce...Ch. 15 - The equilibrium constant Kc for the reaction...Ch. 15 - The dependence of the equilibrium constant of a...Ch. 15 - Prob. 15.147QPCh. 15 - The following diagram shows the variation of the...Ch. 15 - The Kp for the reaction SO2Cl2(g)SO2(g)+Cl2(g) is...Ch. 15 - Derive the equation G=RTlnQK where Q is the...Ch. 15 - Prob. 15.151QPCh. 15 - Prob. 15.152QPCh. 15 - Prob. 15.153QPCh. 15 - Industrial production of ammonia from hydrogen and...Ch. 15 - For which of the following reactions is Kc equal...Ch. 15 - At present, the World Anti-Doping Agency has no...Ch. 15 - (a) Use the vant Hoff equation in Problem 15.146...Ch. 15 - The Ka for hydrocyanic acid (HCN) is 4.9 10 l0....Ch. 15 - Determine the concentrations of Pb2+ and I in a...Ch. 15 - Determine the Ka for a weak acid if a 0.10-M...Ch. 15 - Prob. 15.4KSP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Kc = 5.6 1012 at 500 K for the dissociation of iodine molecules to iodine atoms. I2(g) 2 I(g) A mixture has [I2] = 0.020 mol/Land [I] = 2.0 108 mol/L. Is the reaction at equilibrium (at 500 K)? If not, which way must the reaction proceed to reach equilibrium?arrow_forwardFor which reactions in Exercise 34 is Kp equal to K?arrow_forward12.42 The following reaction is in equilibrium in lake water: HCO,_(aq) + H+(aq)«=*H,CO,(aq) Predict the change in the reaction quotient, Q, for each disturbance below and use that prediction to explain how the equilibrium is shifted by the stress. NaHCOj is added to the lake. H,CO}is added. NaOH is added.arrow_forward
- For the reactionN2O4(g)2NO2(g),Kp=0.25 at a certain temperature. If 0.040 atm of N2O4 is reacted initially, calculate the equilibrium partial pressures of NO2(g) and N2O4(g).arrow_forwardExplain the difference between K, Kp, and Q.arrow_forwardThe following two diagrams represent the composition of an equilibrium mixture for the reaction A2 + B2 2AB at two different temperatures. Based on the diagrams, is the chemical reaction endothermic or exothermic? Explain your answer using Le Chteliers principle. (A atoms are red and B atoms are green in the diagrams.)arrow_forward
- Consider the following reaction at 250C: A(s)+2B(g)C(s)+2D(g) (a) Write an equilibrium constant expression for the reaction. Call the equilibrium constant K1. (b) Write an equilibrium constant expression for the formation of one mole of B(g) and call the equilibrium constant K2. (c) Relate K1 and K2.arrow_forwardFor the equilibrium 2 SO2(g) + O2(g) 2 SO3(g) Kc = 245 (at 1000 K) the equilibrium concentrations are [SO2] = 0.102, [O2] = 0.0132, and [SO3] = 0.184. The concentration of SO2 is suddenly doubled. Show that the forward reaction takes place to reach a new equilibrium.arrow_forwardThe diagram represents an equilibrium mixture for the reaction N2(g) + O2(g) ⇌ 2 NO(g) Estimate the equilibrium constant.arrow_forward
- Phosgene, COCl2, used in the manufacture of polyurethane plastics, is prepared from CO and Cl2. CO(g)+Cl2(g)COCl2(g) An equilibrium mixture at 395C contains 0.012 mol CO and 0.025 mol Cl2 per liter, as well as COCl2 If Kc at 395C is 1.23 103, what is the concentration of COCl2?arrow_forwardA 1.00-L vessel at 400 C contains the following equilibrium concentrations: N2, 1.00 M; H2, 0.50 M; and NH3, 0.25 M. How many moles of hydrogen must be removed from the vessel to increase the concentration of nitrogen to 1.1 M?arrow_forwardFor a typical equilibrium problem, the value of K and the initial reaction conditions are given for a specific reaction, and you are asked to calculate the equilibrium concentrations. Many of these calculations involve solving a quadratic or cubic equation. What can you do to avoid solving a quadratic or cubic equation and still come up with reasonable equilibrium concentrations?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:9781305079113
Author:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:9781305079243
Author:Steven S. Zumdahl, Susan A. Zumdahl
Publisher:Cengage Learning

Chemical Equilibria and Reaction Quotients; Author: Professor Dave Explains;https://www.youtube.com/watch?v=1GiZzCzmO5Q;License: Standard YouTube License, CC-BY