
General, Organic, and Biological Chemistry - 4th edition
4th Edition
ISBN: 9781259883989
Author: by Janice Smith
Publisher: McGraw-Hill Education
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Chapter 15.4, Problem 15.4PP
Interpretation Introduction
(a)
Interpretation:
The enantiomer of given compound that is (R)-limonene should be drawn.
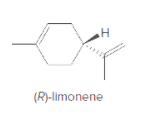
Concept Introduction:
Enantiomers are the compounds having non-superimposable mirror images of each other.
Interpretation Introduction
(b)
Interpretation:
The constitutional isomer of (R)-limonene should be drawn.
Concept Introduction:
Isomers are the compounds having same molecular formula but different structure. The phenomenon possessed by the molecules is known as isomerism.
Compounds having same molecular formula but possessing different connectivity of atoms are known as constitutional isomers.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
4-Chloro-2-pentene has a double bond that can have either the E or the Z configuration and a stereogenic center that can have either the R or the S configuration.
How many stereoisomers arepossible altogether? Draw the structure of each, and group the pairs of enantiomers.
How are the compounds in attached pair related to each other? Are they identical, enantiomers, diastereomers, constitutional isomers, or not isomers of each other??
Identify the stereogenic carbon in (S)- and (R)-limonene, rank the substituents around it and rationalize the assignment of their stereochemical configurations. Hint: When ranking carbons that have multiple bonds, consider the bolded carbon of C=C being connected to 2 carbons and the bolded carbon of C≡C being connected to 3 carbons.
Chapter 15 Solutions
General, Organic, and Biological Chemistry - 4th edition
Ch. 15.1 - Prob. 15.1PCh. 15.1 - Prob. 15.1PPCh. 15.1 - For trans-2-hexene: (a) draw a stereoisomer; (b)...Ch. 15.2 - Prob. 15.3PCh. 15.2 - Prob. 15.4PCh. 15.3 - Prob. 15.2PPCh. 15.3 - Prob. 15.5PCh. 15.3 - Prob. 15.6PCh. 15.3 - Prob. 15.3PPCh. 15.3 - Prob. 15.7P
Ch. 15.3 - Prob. 15.8PCh. 15.3 - Prob. 15.9PCh. 15.4 - Prob. 15.4PPCh. 15.4 - Prob. 15.10PCh. 15.4 - Prob. 15.11PCh. 15.4 - Prob. 15.12PCh. 15.5 - Prob. 15.13PCh. 15.6 - Prob. 15.5PPCh. 15.6 - Prob. 15.14PCh. 15.6 - Prob. 15.15PCh. 15.7 - Prob. 15.16PCh. 15.7 - Prob. 15.17PCh. 15.7 - Prob. 15.6PPCh. 15.7 - Prob. 15.18PCh. 15.8 - Prob. 15.7PPCh. 15.8 - Prob. 15.19PCh. 15.9 - Prob. 15.20PCh. 15 - Prob. 21PCh. 15 - Prob. 22PCh. 15 - Prob. 23PCh. 15 - Prob. 24PCh. 15 - Prob. 25PCh. 15 - Prob. 26PCh. 15 - Prob. 27PCh. 15 - Prob. 28PCh. 15 - Prob. 29PCh. 15 - Prob. 30PCh. 15 - Prob. 31PCh. 15 - Prob. 32PCh. 15 - Prob. 33PCh. 15 - Prob. 34PCh. 15 - Prob. 35PCh. 15 - Prob. 36PCh. 15 - How are the compounds in each pair related? Are...Ch. 15 - Prob. 38PCh. 15 - Prob. 39PCh. 15 - Prob. 40PCh. 15 - Prob. 41PCh. 15 - Prob. 42PCh. 15 - Prob. 43PCh. 15 - Prob. 44PCh. 15 - Prob. 45PCh. 15 - Prob. 46PCh. 15 - Prob. 47PCh. 15 - Prob. 48PCh. 15 - Prob. 49PCh. 15 - Prob. 50PCh. 15 - (a) Define the terms “optically active” and...Ch. 15 - Prob. 52PCh. 15 - Prob. 53PCh. 15 - Prob. 54PCh. 15 - Prob. 55PCh. 15 - Prob. 56PCh. 15 - Prob. 57PCh. 15 - Prob. 58PCh. 15 - Prob. 59PCh. 15 - Prob. 60PCh. 15 - Prob. 61PCh. 15 - Prob. 62PCh. 15 - Prob. 63PCh. 15 - Prob. 64PCh. 15 - Prob. 65PCh. 15 - Prob. 66PCh. 15 - Prob. 67CPCh. 15 - Prob. 68CP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Consider the structure of tallose below and draw the structures of the following: A. its enantiomer B. an epimer of tallose C. alpha-D tallopyranose D. beta-D tallopyranosearrow_forwardDrawn are four isomeric dimethylcyclopropane. a. How are the compounds in each pair related (enantiomers, diastereomers,constitutional isomers): A and B; A and C; B and C; C and D?b. Label each compound as chiral or achiral.c. Which compounds, alone, would be optically active?d. Which compounds have a plane of symmetry?e. Which of the compounds are meso compounds?f. Would an equal mixture of compounds C and D be optically active? Whatabout an equal mixture of B and C?g. How many stereogenic centers are there for each compound?arrow_forward) How many asymmetric carbon atoms are there in an aldotetrose? Draw all thealdotetrose stereoisomersarrow_forward
- Part D. Do the two structures A and B of each pair drawn below represent the same molecule, constitutional isomers, or stereoisomers? If A and B are stereoisomers, further classify them as enantiomers or diastereomers.arrow_forwardLabel compounds B–D as stereoisomers, conformations, or constitutional isomers of Aarrow_forwardIf it is possible, draw its enantiomer, otherwise draw its stereoisomer.arrow_forward
- a.) Draw B as a hexagon with wedges and dashed wedges to show the stereochemistry of substituents.b.) Draw a stereoisomer of A as a hexagon using wedges and dashed wedges to show the orientation of substituents.arrow_forwardDraw all stereoisomers of the following compound and indicate which are enantiomers, disastereomers, or equal molecules.arrow_forwarda. Draw all possible stereoisomers of the following compound: HOCH2CH-OH-CHOH-CH-OHCH2OH b. Which stereoisomers are optically inactive?arrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning


Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning