
Concept explainers
Interpretation: To identify the correct pairing of standard amino acid carbon skeleton degradation product and amino acid genicity.
Concept introduction: Genicity of an amino acid is defined as whether the carbon skeleton degradation product of an amino acid can produce glucose or
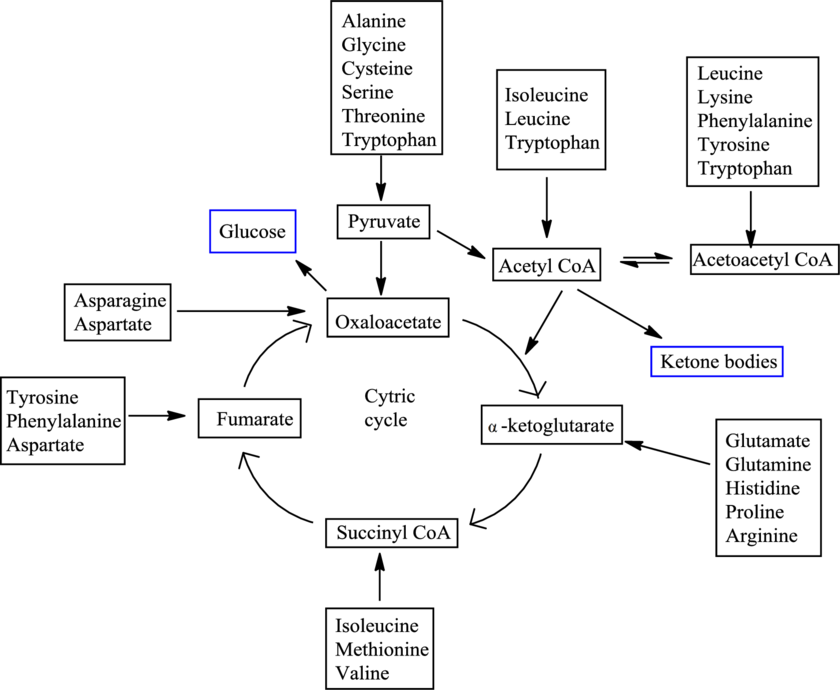
There are 20 standard amino acids and each has a different carbon skeleton. An amino acid is known as a glucogenic amino acid if its carbon-containing degradation product can be used to produce glucose. An amino acid is known as a ketogenic amino acid if its carbon-containing degradation product can be used to produce ketone bodies.
The degradation pathways for different amino acids merge in between and result in the formation of only 7 products. The 7 products are
Fates of carbon skeletons of different amino acids are as follows:
Want to see the full answer?
Check out a sample textbook solution
Chapter 15 Solutions
Organic And Biological Chemistry
- Which of the following statements concerning the degradation of standard amino acid carbon skeletons is correct? a. Each type of carbon skeleton is degraded to a different product. b. All carbon skeletons are degraded to the same product. c. Each type of carbon skeleton is degraded by a different metabolic pathway. d. no correct responsearrow_forwardIn the degradation of heme, the iron atom present is released and then a. eliminated from the body in the urine b. captured by the iron-storage protein ferritin c. captured by the B vitamin niacin d. no correct responsearrow_forwardThe secondary structure of proteins results primarily from which of the following interactions? a. formation of disulfide linkages b. formation of hydrogen bonds c. formation of salt bridges d. more than one response is correct e. no correct response H2 + CH2 = CH2 --> CH3CH3Which of the following could be a substrate for an enzyme that has an induced-fit active site and catalyzes the reaction above?arrow_forward
- Which of the following statements concerning the bile pigments produced from the degradation of the heme portion of hemoglobin is incorrect? a. They are responsible for the yellow color of urine. b. They are responsible for the brown color of feces. c. They are responsible for the yellow color of jaundiced skin. d. no correct responsearrow_forwardQuestion A. Show the ionization equilibrium for the amino acid given and calculate pl. the numbers are the the PKa values B. Predict the organic product if there’s more than one indicate the major C. Which term describes the relationship between the two compounds given a. Enantiomers B. Diasteromers C. Epimers D. Anomers Please answer all questions thank youarrow_forwardA protein has a chain of 200 amino acids. This is the protein's ________ structure a. primary b. secondary c. tertiary d. quaternary The secondary structure of proteins results primarily from which of the following interactions? a. formation of disulfide linkages b. formation of hydrogen bonds c. formation of salt bridges d. more than one response is correct e. no correct response Protein denaturation results in a disruption of the a. primary structure b. secondary and tertiary structure c. primary and secondary structure d. primary, secondary and tertiary structure e. no correct response is given H2 + CH2 = CH2 --> CH3CH3Which of the following could be a substrate for an enzyme that has an induced-fit active site and catalyzes the reaction above? a. CH3_OH b. CH2=CH_CH3 c. CH3_O_CH2CH3 d. none of the above Enzyme activity is best determined by performing a(n) a. enzyme international unit b. enzyme efficiency test c. enzyme specificity test d. enzyme assayarrow_forward
- Which of the following is not an organelle? a. ribosome b. lysosome c. mitochondrion d. no correct responsearrow_forwardIf a mixture of Glu (pI=3.22) and Arg (pI=10.76) is passed through a cation exchange column at pH=7.0, we would expect Glu to elute first and arginine to stick to the column. A. True B. Falsearrow_forward
 Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning

