
Concept explainers
(a)
Interpretation:
The products should be identified by the reaction of 2,6-dimethyl-3-heptanone with
Concept Introduction:
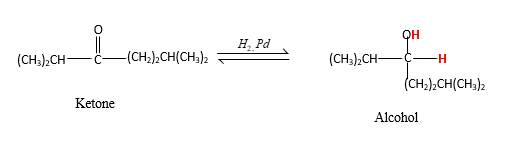
Addition of H2gas to a multiple bond is known as hydrogenation. In the presence of palladium metal as the catalyst, H2 molecules react with
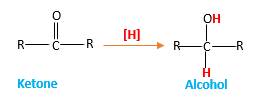
Answer to Problem 78P
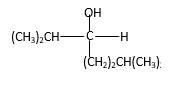
Explanation of Solution
When a ketone reacts with H2 gas in the presence of palladium metal resulting product is the secondary alcohol of the initial ketone molecule. Palladium metal act as a catalyst to the reaction that provides a surface to bind both the H2 and carbonyl compound which reduce the activation energy of the reaction.
Hydrogen atoms in the alcohol molecule shown below, which are indicated in red color are the added H during the hydrogenation reaction.
(b)
Interpretation:
The products should be identified by the reaction of 2,6-dimethyl-3-heptanone with
Concept Introduction:
Addition of an O atom in to a molecule is known as oxidation. If a carbonyl atom consists of a hydrogen atom directly connected to the carbonyl C, it will be oxidized in the presence of an oxidizing agent such as
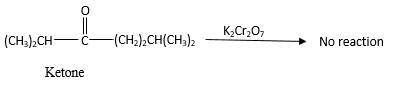
Answer to Problem 78P
No reaction.
Explanation of Solution
Addition of an O atom in to a molecule is known as oxidation. If a carbonyl atom consists of a hydrogen atom directly connected to the carbonyl C, it will be oxidized in the presence of an oxidizing agent such as
Hence, during the reaction no color change can be observed.
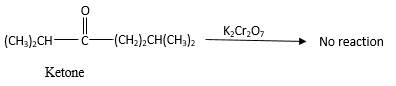
(c)
Interpretation:
The products should be identified by the reaction of 2,6-dimethyl-3-heptanone with
Concept Introduction:
Addition of an O atom in to a molecule is known as oxidation. If a carbonyl atom consists of a hydrogen atom directly connected to the carbonyl C, it will be oxidized in the presence of an oxidizing agent such as (
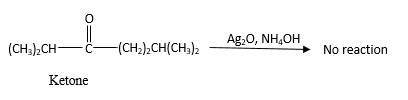
Answer to Problem 78P
No reaction.
Explanation of Solution
Addition of an O atom in to a molecule is known as oxidation. If a carbonyl atom consists of a hydrogen atom directly connected to the carbonyl C in an it will be oxidized in the presence of an oxidizing agent such as
Hence, during the reaction is no silver mirror can be observed.
(d)
Interpretation:
The products should be identified by the reaction of 2,6-dimethyl-3-heptanone with
Concept Introduction:
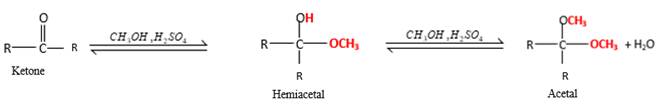
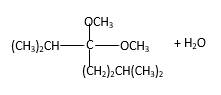
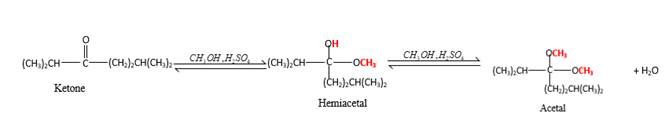
In the presence of alcohol in the acidic medium, ketones undergo addition reactions and give acetal in two steps.
Hydrogen atom and CH3 groups in the acetal and hemiacetal molecules shown below, which are indicated in red color are the added molecules during the reaction.
Answer to Problem 78P
Explanation of Solution
In the presence of alcohol in the acidic medium, ketones undergo addition reactions and give acetal in two steps. In the first step ketones form hemiacetals and during the second step it converts to an acetal molecule of the respective ketone molecule.
Addition of one molecule of alcohol in to a ketone forms a hemiacetal, one bond of the
Hydrogen atom, CH3 and OCH3 groups in the acetal and hemiacetal molecules shown below, which are indicated in red color are the added molecules during the reaction.
(e)
Interpretation:
The products should be identified by the reaction of 2,6-dimethyl-3-heptanone with
Concept Introduction:
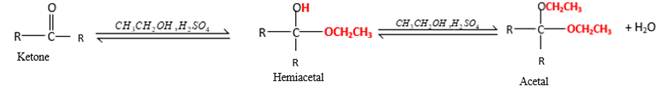
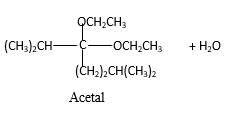
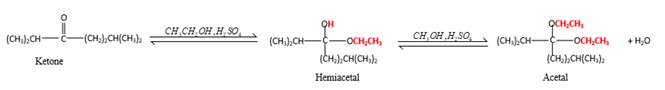
In the presence of alcohol in the acidic medium,
Hydrogen atom and CH2CH3 groups in the acetal and hemiacetal molecules shown below, which are indicated in red color are the added molecules during the reaction.
Answer to Problem 78P
Explanation of Solution
In the presence of alcohol in the acidic medium, ketones undergo addition reactions and give acetal in two steps. In the first step ketones form hemiacetals and during the second step it converts to an acetal molecule of the respective ketone molecule.
Addition of one molecule of alcohol in to aketone forms a hemiacetal, one bond of the
Hydrogen atom, CH2CH3 and OCH2CH3 groups in the acetal and hemiacetal molecules shown below, which are indicated in red color are the added molecules during the reaction.
(f)
Interpretation:
The products should be identified by the reaction of
Concept Introduction:
In the presence of water and acid, acetals undergo hydrolysis reaction and produce aldehydes.
OR' groups in the acetal molecule shown below, which are indicated in red color are the molecules which becomes alcohol molecules during the hydrolysis.
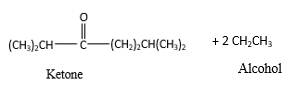
Answer to Problem 78P
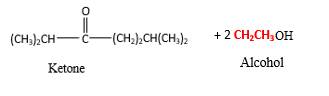
Explanation of Solution
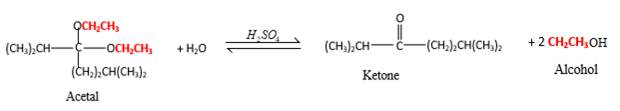
Acetals are stable molecules, but their bonds can cleave by a reaction with water and produce aldehydes.
In the acetal molecule, two bonds of the
CH2CH3 groups in the acetal molecule shown below, which are indicated in red color are the molecules which becomes alcohol molecules during the hydrolysis.
Want to see more full solutions like this?
Chapter 16 Solutions
General, Organic, and Biological Chemistry - 4th edition
- What is the product of the reaction of 2-methyl-2-butene with Br2/H2O? 3-bromo-2-methyl-2-butanol 2-bromo-3-methyl-2-butanol 4-bromo-2-methyl-2-butanol 2,3-dibromo-2-methylbutanearrow_forwardDraw the products formed when ethylene oxide is treated with each reagent. a. HBr b. H2O(H2SO4) c. [1] CH3CH2O; [2] H2O d. [1] HC ≡ C−; [2] H2O e. [1] −OH; [2] H2O f. [1] CH3S−; [2] H2Oarrow_forward28. General formula of AZO compounds.a. R-N=N-Ra. R-N≣Nb. R-NH3c. none of these 29. Which is the nitro group?a. -NH3b. -NO2c. -C≣Nd. none of these 30. General formula of sulfones:a. R-OSRb. R-SHc. R-S-Rd. R-SO2arrow_forward
- Draw the products formed when phenol(C6H5OH) is treated with each reagent. Give an explanation. c. CH3CH2Cl, AlCl3 l. product in (c), then KMnO4arrow_forwardShow how to prepare each compound from 2-methyl- 1- propanol. a. 2- methylpropene b. 2- methyl- 2- propanol c. 2- methylpropanoic acid (CH3)2CHCOOHarrow_forward"A research team synthesizes a novel organic compound 'X' with the molecular formula C5H8O2. When 'X' is treated with a deuterated acid (D2O), a single deuterium atom replaces a hydrogen atom, forming compound 'Y' (C5H7DO2). 'X' does not react with 2,4-Dinitrophenylhydrazine (2,4-DNP) but does react with both Tollens' reagent and Benedict's solution, forming a silver mirror and a red precipitate, respectively. Furthermore, 'X' undergoes catalytic hydrogenation over a palladium catalyst, consuming one mole of hydrogen to form a compound 'Z' (C5H10O2). Based on these observations, what is the most likely structure of compound 'X'?" A. Methyl vinyl ketone B. 3-Buten-2-one C. Acetoacetic ester D. 2-Hydroxypent-3-enalarrow_forward
- 1a. what would be the theoretical yield of 2-chloro-2methyl-butane using 5 ml of 2-methyl-2-butanol and 10 ml of HCL or HBr? 1b. what would be the theoretical yield of 2-bromo-2methyl-butane using 5 ml of 2-methyl-2-butanol and 10 ml of HCL or HBr?arrow_forwardMolecule Type Boiling point (°C) CH3CH2CH3 Alkane -42 CH3CHO Aldehyde +21 CH3CH2OH Alcohol +78 i. Why is the boiling point of the aldehyde greater than that of the alkane?ii. Why is the boiling point of alcohol the highest?iii. Explain why the solubility of aldehydes and alcohols falls as the molecules get bigger.arrow_forward1. what priority functional group of ff organic compound? a. carboxyl b. hydroxide c. hydroxyl d. carbonyl 2. what group does the ff organic compound belong? a. azo b. diazo c. aromatic d. organosulfur 3. what group does the ff organic compound belong? a. azo b. amines c. diazo d. nitrilesarrow_forward
- 1. What is the role of the acetic acid in the oxidation of Cyclohexanol to Cyclohexanone? Write the balanced chemical reaction between acetic acid and sodium hypochlorite.2. How do you neutralize the acetic acid regenerated in the reaction? Write the balanced chemical reaction.arrow_forwardWhich of the following alcohols can be prepared from a Grignard reagent and ethylene oxide? A. only 1 B. only 1 and 2 C. only 1, 2 and 3 D. 1, 2, 3 and 4arrow_forward#20 B Draw structural formulas for all possible carbocations formed by the reaction of each alkene with HCl.arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

