
Concept explainers
a)
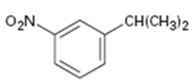
Interpretation:
The
Concept introduction:
The benzene ring is inert to even strong oxidizing agents like KMnO4 and Na2Cr2O7. However the alkyl side chains with benzylic hydrogen atoms are readily oxidized by these reagents into carboxyl groups. Compounds having no benzylic hydrogen atoms in the side chain are not oxidized.
To give:
The aromatic substance obtained as the product, when m-nitroisopropyl benzene is oxidized with KMnO4.
b)
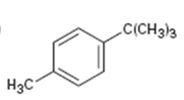
Interpretation:
The aromatic substance obtained as the product, when p-tert-butyltoluene is oxidized with KMnO4, is to be given.
Concept introduction:
The benzene ring is inert to even strong oxidizing agents like KMnO4 and Na2Cr2O7. However the alkyl side chains with benzylic hydrogen atoms are readily oxidized by these reagents into carboxyl groups. Compounds having no benzylic hydrogen atoms in the side chain are not oxidized.
To explain:
The aromatic substance obtained as the product when p-tert-butyl toluene is oxidized with KMnO4.
Trending nowThis is a popular solution!

Chapter 16 Solutions
Organic Chemistry
- Propose structures for hydrocarbons that give the following products on oxidative cleavage by KMnO4 or O3:arrow_forwardPredict the products of NaBH4 reduction of the following compounds?arrow_forwardShow how would you synthesize the following aromatic derivatives from benzene.i) p-t-butylnitrobenzene ii) p-toluene sulfonic acid iii) p-chloro toluenearrow_forward
- Show how you would synthesize the following aromatic derivatives from benzene.(a) p-tert-butylnitrobenzenearrow_forwardHow would you accomplish the following conversion of nitrobenzene to benzene?arrow_forwardWhat products will be obtained from the E1 reaction of the alkyl halides in Problem 45?arrow_forward
- Which of the reactions below are correctly identified? These are SN2 reactions:arrow_forwardStarting from any aromatic hydrocarbon of your choice, how would you synthesize the following substances? Ortho and para isomers can be separated if necessary.arrow_forwardPropose a structure for an aromatic hydrocarbon, C10H14, that can form only one C10H13Cl product on substitution of a hydrogen on the aromatic ring with chlorine.arrow_forward
- What would the major organic reaction product be from the reaction of 1-bromo-1-methylcyclopentane withsodium hydroxide? Would the elimination reaction outcome be affected if a student accidentally adds sodium tertbutoxide instead of sodium hydroxide?arrow_forwardAlkenyl halides such as vinyl bromide, CH2=CHBr, undergo neither SN1 nor SN2 reactions. What factors account for this lack of reactivity?arrow_forwardWhat condensation product would you expect to obtain by treatment of the following substances with sodium ethoxide in ethanol. Cycloheptanonearrow_forward
