
Interpretation:
A mechanism for the reaction of benzene with 2,2,5,5-teteamethyltetrahydrofuran shown below is to be proposed.
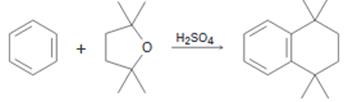
Concept introduction:
The reaction involves the electrophilc substitution of cyclic ether into a benzene ring. The electrophile can be produced by the protonation of the ethereal oxygen followed by a cleavage of a C-O bond. The eletrophilic attack will lead to the formation of an alcohol. Protonation of the alcohol and subsequent elimination of water generates another carbocation which can internally attack the ring. The loss of a proton from the intermediate will lead to product expected.
To propose:
A mechanism for the reaction of benzene with 2,2,5,5-teteamethyltetrahydrofuran shown.
Trending nowThis is a popular solution!

Chapter 16 Solutions
Organic Chemistry
- Acetoxybenzene (PhOC(=O)OCH3) is much less reactive than ethoxybenzene (PhOCH2CH3) in electrophilic aromatic substitution reactions. Suggest an explanation for this result, based on an analysis of the inductive and resonance electronic effects of the two substituents on the stability of theWheland intermediate for para substitution by an electrophile E+arrow_forwardbenzene to p-bromobenzoic synthesisarrow_forwardDraw and explain the mechanism of substitution of 3-bromopentane in the presence of ammonia. explain clearly.arrow_forward
- Give only typing answer with explanation and conclusion to all parts draw the mechanism for these 4 reactions with electron movement and arrows and products formed The dehydration of 2-butanol with H2SO4 The dehydration of 2-butanol with H2SO4 The dehydrobromination of 1-bromobutane with potassium tert-butoxide The dehydrobromination of 2-bromobutane with potassium tert-butoxidearrow_forwardChemistry Q: Kindly explain the striking observation that methylation of the alcohol 1 leads to a methoxy compound in which the stereochemistry of the oxygen has changed. Explain it by providing a mechanism accounting for the transformation. (write legibly pls).arrow_forwardSynthesize from benzene. (Hint: All of these require diazonium ions.) ) 4-methoxyanilinearrow_forward
- Complete the following mechanism for the acid-catalyzed rearrangement of 1-vinyl-2-phenylcyclopropane to give both isomers (E / Z) of 5-bromo-5-phenyl-2-pentene employing the curved-arrow/arrow-pushing formalism.arrow_forwardIllustrate the mechanism of forming alkynes from a 1,2-dihalide and a vinylichalide. Show intermediates as applicable and use the arrow formalisms.arrow_forwardHow many alkenes yield 2,2,3,4,4−pentamethylpentane on catalytic hydrogenation?arrow_forward
- Write the mechanism for the following reactions. Explain detail each steps of reactions.a) Halogenation of benzene b) Nitration of benzene c) Sulfonation of benzenearrow_forwardWrite down all possible alkene products from the following Elemination reaction (no mechanism is required) indicate which one will be major product. And explain it?arrow_forwardplease explain clearly and in detail. thank you! 2-butene reacts with NBS yields and forms two products, 1-bromobut-2-ene and 3-bromobut-1-ene. explain how this happens using mechanisms.arrow_forward

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

