
Chemistry: An Atoms First Approach
2nd Edition
ISBN: 9781305079243
Author: Steven S. Zumdahl, Susan A. Zumdahl
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 17, Problem 10ALQ
What is the difference between 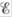 and
and 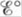 ? When is
? When is 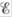 equal to zero? When is
equal to zero? When is 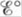 equal to zero? (Consider “regular” galvanic cells as well as concentration cells.)
equal to zero? (Consider “regular” galvanic cells as well as concentration cells.)
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Chapter 17 Solutions
Chemistry: An Atoms First Approach
Ch. 17 - What is a half-reaction? Why must the number of...Ch. 17 - Galvanic cells harness spontaneous...Ch. 17 - Table 17-1 lists common half-reactions along with...Ch. 17 - Prob. 4RQCh. 17 - The Nernst equation allows determination of the...Ch. 17 - Prob. 6RQCh. 17 - Prob. 7RQCh. 17 - Prob. 8RQCh. 17 - What characterizes an electrolytic cell? What is...Ch. 17 - Sketch a galvanic cell, and explain how it works....
Ch. 17 - Prob. 2ALQCh. 17 - Prob. 3ALQCh. 17 - Prob. 4ALQCh. 17 - Sketch a cell that forms iron metal from iron(II)...Ch. 17 - Which of the following is the best reducing agent:...Ch. 17 - Prob. 7ALQCh. 17 - Prob. 8ALQCh. 17 - Explain why cell potentials are not multiplied by...Ch. 17 - What is the difference between and ? When is equal...Ch. 17 - Prob. 11ALQCh. 17 - Look up the reduction potential for Fe3+ to Fe2+....Ch. 17 - Prob. 13ALQCh. 17 - Is the following statement true or false?...Ch. 17 - Prob. 15RORRCh. 17 - Assign oxidation numbers to all the atoms in each...Ch. 17 - Specify which of the following equations represent...Ch. 17 - The Ostwald process for the commercial production...Ch. 17 - Prob. 19QCh. 17 - Prob. 20QCh. 17 - When magnesium metal is added to a beaker of...Ch. 17 - How can one construct a galvanic cell from two...Ch. 17 - The free energy change for a reaction, G, is an...Ch. 17 - What is wrong with the following statement: The...Ch. 17 - When jump-starting a car with a dead battery, the...Ch. 17 - Prob. 26QCh. 17 - Prob. 27QCh. 17 - Consider the following electrochemical cell: a. If...Ch. 17 - Balance the following oxidationreduction reactions...Ch. 17 - Prob. 30ECh. 17 - Prob. 31ECh. 17 - Prob. 32ECh. 17 - Chlorine gas was first prepared in 1774 by C. W....Ch. 17 - Gold metal will not dissolve in either...Ch. 17 - Prob. 35ECh. 17 - Consider the following galvanic cell: a. Label the...Ch. 17 - Prob. 37ECh. 17 - Sketch the galvanic cells based on the following...Ch. 17 - Prob. 39ECh. 17 - Prob. 40ECh. 17 - Prob. 41ECh. 17 - Prob. 42ECh. 17 - Prob. 43ECh. 17 - Give the standard line notation for each cell in...Ch. 17 - Prob. 45ECh. 17 - Prob. 46ECh. 17 - Prob. 47ECh. 17 - Prob. 48ECh. 17 - Prob. 49ECh. 17 - The amount of manganese in steel is determined by...Ch. 17 - Prob. 51ECh. 17 - Prob. 52ECh. 17 - Estimate for the half-reaction 2H2O+2eH2+2OH given...Ch. 17 - Prob. 54ECh. 17 - Glucose is the major fuel for most living cells....Ch. 17 - Direct methanol fuel cells (DMFCs) have shown some...Ch. 17 - Prob. 57ECh. 17 - Using data from Table 17-1, place the following in...Ch. 17 - Answer the following questions using data from...Ch. 17 - Prob. 60ECh. 17 - Consider only the species (at standard conditions)...Ch. 17 - Prob. 62ECh. 17 - Prob. 63ECh. 17 - Prob. 64ECh. 17 - Prob. 65ECh. 17 - Prob. 66ECh. 17 - A galvanic cell is based on the following...Ch. 17 - Prob. 68ECh. 17 - Consider the concentration cell shown below....Ch. 17 - Prob. 70ECh. 17 - The overall reaction in the lead storage battery...Ch. 17 - Prob. 72ECh. 17 - Consider the cell described below:...Ch. 17 - Consider the cell described below:...Ch. 17 - Prob. 75ECh. 17 - Prob. 76ECh. 17 - Prob. 77ECh. 17 - Prob. 78ECh. 17 - Prob. 79ECh. 17 - An electrochemical cell consists of a nickel metal...Ch. 17 - An electrochemical cell consists of a standard...Ch. 17 - Prob. 82ECh. 17 - Consider a concentration cell that has both...Ch. 17 - Prob. 84ECh. 17 - Prob. 85ECh. 17 - Prob. 86ECh. 17 - Consider the following galvanic cell at 25C:...Ch. 17 - Prob. 88ECh. 17 - Prob. 89ECh. 17 - Prob. 90ECh. 17 - Prob. 91ECh. 17 - The solubility product for CuI(s) is 1.1 102...Ch. 17 - How long will it take to plate out each of the...Ch. 17 - The electrolysis of BiO+ produces pure bismuth....Ch. 17 - What mass of each of the following substances can...Ch. 17 - Prob. 96ECh. 17 - An unknown metal M is electrolyzed. It took 74.1 s...Ch. 17 - Electrolysis of an alkaline earth metal chloride...Ch. 17 - What volume of F2 gas, at 25C and 1.00 atm, is...Ch. 17 - What volumes of H2(g) and O2(g) at STP are...Ch. 17 - Prob. 101ECh. 17 - A factory wants to produce 1.00 103 kg barium...Ch. 17 - It took 2.30 min using a current of 2.00 A to...Ch. 17 - A solution containing Pt4+ is electrolyzed with a...Ch. 17 - A solution at 25C contains 1.0 M Cd2+, 1.0 M Ag+,...Ch. 17 - Consider the following half-reactions: A...Ch. 17 - In the electrolysis of an aqueous solution of...Ch. 17 - Copper can be plated onto a spoon by placing the...Ch. 17 - Prob. 109ECh. 17 - Prob. 110ECh. 17 - Prob. 111ECh. 17 - What reaction will take place at the Cathode and...Ch. 17 - Gold is produced electrochemically from an aqueous...Ch. 17 - Prob. 114AECh. 17 - The saturated calomel electrode. abbreviated SCE....Ch. 17 - Consider the following half-reactions: Explain why...Ch. 17 - Consider the standard galvanic cell based on the...Ch. 17 - Prob. 118AECh. 17 - The black silver sulfide discoloration of...Ch. 17 - Prob. 120AECh. 17 - When aluminum foil is placed in hydrochloric acid,...Ch. 17 - Prob. 122AECh. 17 - Prob. 123AECh. 17 - The overall reaction and equilibrium constant...Ch. 17 - What is the maximum work that can be obtained from...Ch. 17 - The overall reaction and standard cell potential...Ch. 17 - Prob. 127AECh. 17 - Prob. 128AECh. 17 - Prob. 129AECh. 17 - Prob. 130AECh. 17 - Prob. 131AECh. 17 - Prob. 132AECh. 17 - Prob. 133AECh. 17 - Prob. 134CWPCh. 17 - Consider a galvanic cell based on the following...Ch. 17 - Consider a galvanic cell based on the following...Ch. 17 - Consider a galvanic cell based on the following...Ch. 17 - An electrochemical cell consists of a silver metal...Ch. 17 - An aqueous solution of PdCl2 is electrolyzed for...Ch. 17 - Prob. 140CPCh. 17 - Prob. 141CPCh. 17 - The overall reaction in the lead storage battery...Ch. 17 - Consider the following galvanic cell: Calculate...Ch. 17 - Prob. 144CPCh. 17 - A galvanic cell is based on the following...Ch. 17 - Prob. 146CPCh. 17 - The measurement of pH using a glass electrode...Ch. 17 - Prob. 148CPCh. 17 - A galvanic cell is based on the following...Ch. 17 - Prob. 150CPCh. 17 - Prob. 151CPCh. 17 - Prob. 152CPCh. 17 - Consider the following galvanic cell: A 15 0-mole...Ch. 17 - When copper reacts with nitric acid, a mixture of...Ch. 17 - The following standard reduction potentials have...Ch. 17 - An electrochemical cell is set up using the...Ch. 17 - Three electrochemical cells were connected in...Ch. 17 - A silver concentration cell is set up at 25C as...Ch. 17 - A galvanic cell is based on the following...Ch. 17 - Prob. 160MP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Is it reasonable to conclude that a potential could be assigned to each half-cell in a voltaic cell, based on these data for three voltaic cells? Explain. Zn(s)|Zn2+(aq)||Cu2+(aq)|Cu(s) cell potential=1.10 V Zn(s)|Zn2+(aq)||Ag+(aq)|Ag(s) cell potential=1.56 V Cu(s)|Cu2+(aq)||Ag+(aq)|Ag(s) cell potential=0.46 Varrow_forwardDetermine the overall reaction and its standard cell potential at 25 C for this reaction. Is the reaction spontaneous at standard conditions? Cu(s)|Cu2+(aq)Au3+(aq)|Au(s)arrow_forwardWhat is the voltage of a concentration cell of Cl ions where the concentrations are 1.045 and 0.085 M? What the spontaneous reaction?arrow_forward
- Use the following half-equations to write three spontaneous reactions. Justify your answers by calculating E° for the cells. 1. MnO4(aq)+8H+(aq)+5eMn2+(aq)+4H2OE=+1.512V 2. O2(g)+4H+(aq)+4e2H2OE=+1.229V 3. Co2+(aq)+2eCo(s)E=0.282Varrow_forwardIs the following statement true or false? Concentration cells work because standard reduction potentials are dependent on concentration. Explain.arrow_forwardConsider a cell reaction at 25°C where n=4 . Fill in the following table.arrow_forward
- For each reaction listed, determine its standard cell potential at 25 C and whether the reaction is spontaneous at standard conditions. (a) Mn(s)+Ni2+(aq)Mn2+(aq)+Ni(s) (b) 3Cu2+(aq)+2Al(s)2Al3+(aq)+3Cu(s) (c) Na(s)+LiNO3(aq)NaNO3(aq)+Li(s) (d) Ca(NO3)2(aq)+Ba(s)Ba(NO3)2(aq)+Ca(s)arrow_forwardIf the cell potential is proportional to work and the standard reduction potential for the hydrogen ion is zero, does this mean that the reduction of the hydrogen ion requires no work?arrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:9781305079243
Author:Steven S. Zumdahl, Susan A. Zumdahl
Publisher:Cengage Learning


Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning


Principles of Modern Chemistry
Chemistry
ISBN:9781305079113
Author:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:Cengage Learning
Introduction to Electrochemistry; Author: Tyler DeWitt;https://www.youtube.com/watch?v=teTkvUtW4SA;License: Standard YouTube License, CC-BY