
Concept explainers
The overall reaction and equilibrium constant value for a hydrogen-oxygen fuel cell at 298 K is
a. Calculate 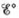 and ∆G° at 298 K for the fuel cell reaction.
and ∆G° at 298 K for the fuel cell reaction.
b. Predict the signs of ∆H° and ∆S° for the fuel cell reaction.
c. As temperature increases, does the maximum amount of work obtained from the fuel cell reaction increase, decrease, or remain the same? Explain.
Trending nowThis is a popular solution!

Chapter 17 Solutions
Chemistry: An Atoms First Approach
Additional Science Textbook Solutions
Organic Chemistry
Chemistry by OpenStax (2015-05-04)
Organic Chemistry - Standalone book
Fundamentals of Heat and Mass Transfer
Chemistry: The Central Science (13th Edition)
- Calculate the standard cell potential of the cell corresponding to the oxidation of oxalic acid, H2C2O4, by permanganate ion. MnO4. 5H2C2O4(aq)+2MnO4(aq)+6H+(aq)10CO2(g)+2Mn2+(aq)+8H2O(l) See Appendix C for free energies of formation: Gf for H2C2O4(aq) is 698 kJ.arrow_forwardA voltaic cell is constructed in which one half-cell consists of a silver wire in an aqueous solution of AgNO3.The other half cell consists of an inert platinum wire in an aqueous solution containing Fe2+(aq) and Fe3+(aq). (a) Calculate the cell potential, assuming standard conditions. (b) Write the net ionic equation for the reaction occurring in the cell. (c) Which electrode is the anode and which is the cathode? (d) If [Ag+] is 0.10 M, and [Fe2+] and [Fe3+] are both 1.0 M, what is the cell potential? Is the net cell reaction still that used in part (a)? If not, what is the net reaction under the new conditions?arrow_forwardAn electrode is prepared from liquid mercury in contact with a saturated solution of mercury(I) chloride, Hg2Cl, containing 1.00 M Cl . The cell potential of the voltaic cell constructed by connecting this electrode as the cathode to the standard hydrogen half-cell as the anode is 0.268 V. What is the solubility product of mercury(I) chloride?arrow_forward
- Calculate the cell potential of a cell operating with the following reaction at 25C, in which [MnO4] = 0.010 M, [Br] = 0.010 M. [Mn2] = 0.15 M, and [H] = 1.0 M. 2MNO4(aq)+10Br(aq)+16H+(aq)2MN2(aq)+5Br2(l)+8H2O(l)arrow_forwardAn electrolysis experiment is performed to determine the value of the Faraday constant (number of coulombs per mole of electrons). In this experiment, 28.8 g of gold is plated out from a AuCN solution by running an electrolytic cell for two hours with a current of 2.00 A. What is the experimental value obtained for the Faraday Constant?arrow_forwardCalculate the cell potential of a cell operating with the following reaction at 25C, in which [Cr2O32] = 0.020 M, [I] = 0.015 M, [Cr3+] = 0.40 M, and [H+] = 0.60 M. Cr2O72(aq)+6I(aq)+14H+(aq)2Cr3+(aq)+3I2(s)+7H2O(l)arrow_forward
- Assume the following electrochemical cell simulates the galvanic cell formed by copper and zinc in seawater at pH 7.90 and 25 C. Zn | Zn(OH)2(s) | OH(aq) || Cu(OH)2(s) | Cu(s) a. Write a balanced equation for the reaction that occurs at the cathode. b. Write a balanced equation for the reaction that occurs at the anode. c. Write a balanced chemical equation for the overall reaction. d. Determine the potential (in volts) of the cell.arrow_forwardWhat is the standard cell potential you would obtain from a cell at 25C using an electrode in which Hg22+(aq) is in contact with mercury metal and an electrode in which an aluminum strip dips into a solution of Al3+(aq)?arrow_forwardConsider the electrolysis of water in the presence of very dilute H2SO4. What species is produced at the anode? Atthe cathode? What are the relative amounts of the speciesproduced at the two electrodes?arrow_forward
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning





