
A galvanic cell is based on the following half-reactions:
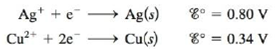
In this cell, the silver compartment contains a silver electrode and excess AgCl(s) (Ksp = 1.6 × 10−10), and the copper compartment contains a copper electrode and [Cu2+] = 2.0 M.
a. Calculate the potential for this cell at 25°C.
b. Assuming 1.0 L of 2.0 M Cu2+ in the copper compartment, calculate the moles of NH3 that would have to be added to give a cell potential of 0.52 Vat 25°C (assume no volume change on addition of NH3).
Trending nowThis is a popular solution!

Chapter 17 Solutions
Chemistry: An Atoms First Approach
Additional Science Textbook Solutions
Organic Chemistry As a Second Language: Second Semester Topics
General Chemistry: Principles and Modern Applications (11th Edition)
Elementary Principles of Chemical Processes, Binder Ready Version
General, Organic, and Biological Chemistry - 4th edition
- An electrochemical cell consists of a nickel metal electrode immersed in a solution with [Ni2+] = 1.0 M separated by a porous disk from an aluminum metal electrode. a. What is the potential of this cell at 25C if the aluminum electrode is placed in a solution in which [Al3+] = 7.2 103 M? b. When the aluminum electrode is placed in a certain solution in which [Al3+] is unknown, the measured cell potential at 25C is 1.62 V. Calculate [Al3+] in the unknown solution. (Assume Al is oxidized.)arrow_forwardConsider a galvanic cell based on the following half-reactions: a. What is the standard potential for this cell? b. A nonstandard cell is set up at 25C with [Mg2+] = 1.00 105 M. The cell potential is observed to be 4.01 V. Calculate [Au3+] in this cell.arrow_forwardA galvanic cell is based on the following half-reactions: In this cell, the copper compartment contains a copper electrode and [Cu2+] = 1.00 M, and the vanadium compartment contains a vanadium electrode and V2+ at an unknown concentration. The compartment containing the vanadium (1.00 L of solution) was titrated with 0.0800 M H2EDTA2, resulting in the reaction H2EDTA2(aq)+V2+(aq)VEDTA2(aq)+2H+(aq)K=? The potential of the cell was monitored to determine the stoichiometric point for the process, which occurred at a volume of 500.0 mL H2EDTA2 solution added. At the stoichiometric point, was observed to be 1 .98 V. The solution was buffered at a pH of 10.00. a. Calculate before the titration was carried out. b. Calculate the value of the equilibrium constant, K, for the titration reaction. c. Calculate at the halfway point in the titration.arrow_forward
- Consider a galvanic cell based on the following half-reactions: a. What is the expected cell potential with all components in their standard states? b. What is the oxidizing agent in the overall cell reaction? c. What substances make up the anode compartment? d. In the standard cell, in which direction do the electrons flow? e. How many electrons are transferred per unit of cell reaction? f. If this cell is set up at 25C with [Fe2+] = 2.00 104 M and [La3+] = 3.00 103 M, what is the expected cell potential?arrow_forwardCalculate the cell potential of a cell operating with the following reaction at 25C, in which [MnO4] = 0.010 M, [Br] = 0.010 M. [Mn2] = 0.15 M, and [H] = 1.0 M. 2MNO4(aq)+10Br(aq)+16H+(aq)2MN2(aq)+5Br2(l)+8H2O(l)arrow_forwardAn aqueous solution of an unknown salt of gold is electrolyzed by a current of 2.75 amps for 3.39 hours. The electroplating is carried out with an efficiency of 93.0%, resulting in a deposit of 21.221 g of gold. a How many faradays are required to deposit the gold? b What is the charge on the gold ions (based on your calculations)?arrow_forward
- An electrolysis experiment is performed to determine the value of the Faraday constant (number of coulombs per mole of electrons). In this experiment, 28.8 g of gold is plated out from a AuCN solution by running an electrolytic cell for two hours with a current of 2.00 A. What is the experimental value obtained for the Faraday Constant?arrow_forwardAn electrochemical cell consists of a silver metal electrode immersed in a solution with [Ag+] = 1.0 M separated by a porous disk from a copper metal electrode. If the copper electrode is placed in a solution of 5.0 M NH3 that is also 0.010 M in Cu(NH3)42+, what is the cell potential at 25C? Cu2+(aq)+4NH3(aq)Cu(NH3)42+(aq)K=1.01013arrow_forwardGiven this reaction, its standard potential, and the standard half-cell potential of 0.34 V for the Cu2+ |Cu half-cell, calculate E° for the Fe(s)|Fe2+(aq) half-cell.arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
 Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning





